Playlist
Show Playlist
Hide Playlist
Carbamoyl Phosphate Synthetase Reaction and Citrulline
-
Slides UreaCycle Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
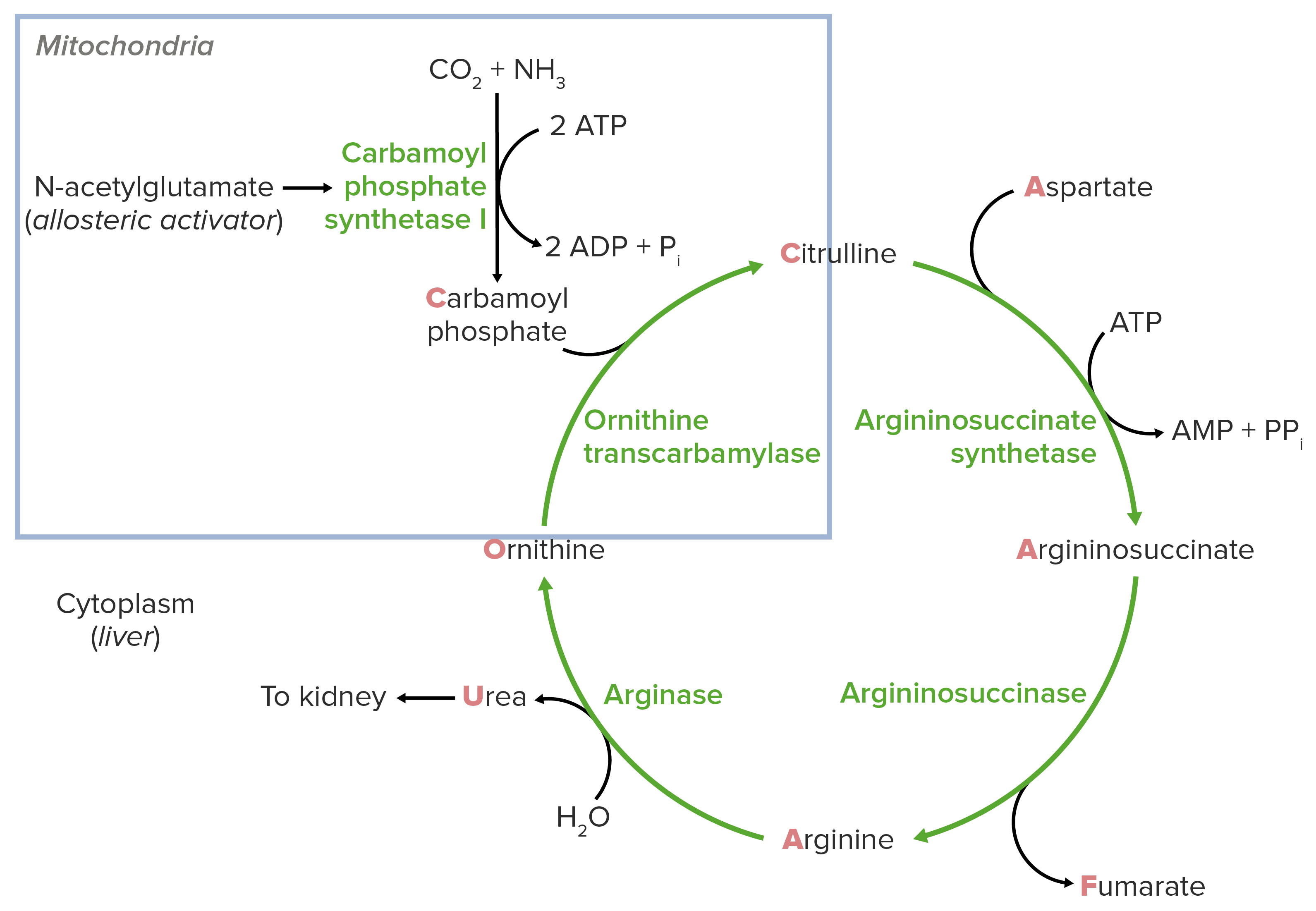
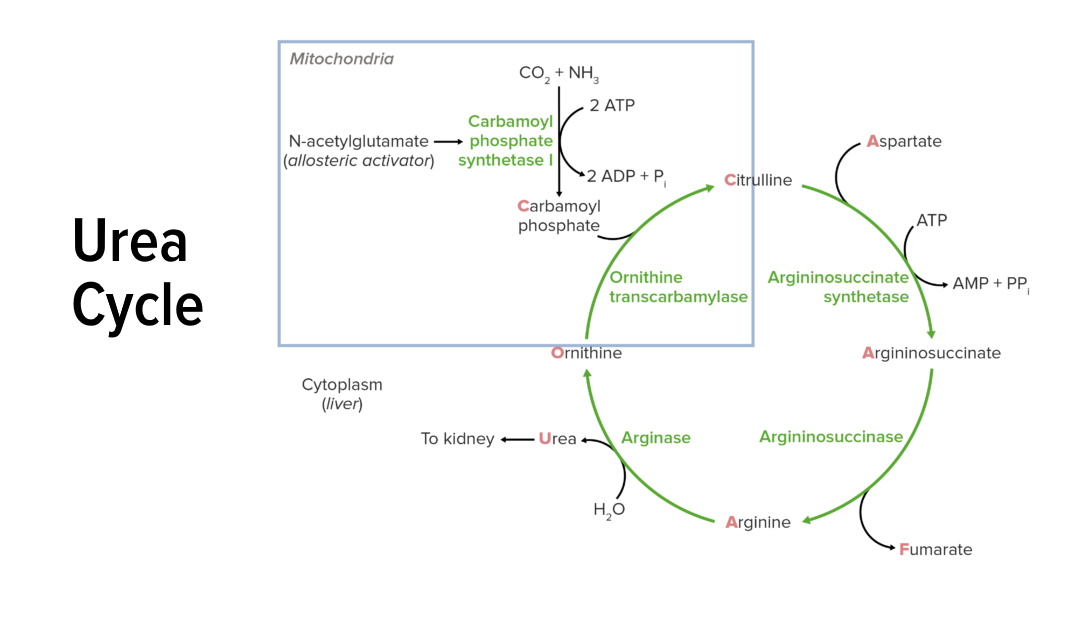
00:02 So I've now gone through the cycle as a whole. 00:04 What I want to do now is focus in a little bit more closely on each of the enzymes that I've been talking about. 00:09 The first enzyme of course is carbamoyl phosphate synthetase. 00:13 Now, this reaction requires ammonium ion. 00:15 And the ammonium ion in this reaction is ultimately coming from glutamine or glutamate. 00:21 These may have gotten their ammine from another amino acid. 00:26 The reaction requires the action of either glutaminase in the case of breaking down glutamine to make ammonium ion or glutamate dehydrogenase which breaks down glutamate to get that ammonium ion. 00:37 We see these reactions here. 00:39 It's a hydrolysis in the case of glutamine to release and produce glutamate plus that ammonium, or it's also hydrolysis in the case of glutamate that produces alpha-ketoglutarate and the ammonium ion as we see here. 00:51 In the case of the glutamate dehydrogenase that's an oxidation as the name dehydrogenase would suggest and the electrons are donated to NADP to make NAPH. 01:01 So, we have produced the ammonia or ammonium ion necessary for the reaction I've described. 01:06 The production of the bicarbonate is done very easily within the cell and I won't go into that right here. 01:11 We thus have the bicarbonate and the ammonium that we need for the reaction catalyzed by the carbamoyl of phosphate synthethase and we see that happening right here coming either from the glutamine side or from the glutamate side. 01:24 The net result is that we have made the carbamoyl phosphate necessary for the feeder reaction. 01:32 The ornithine transcarbamoylase reaction is shown here. 01:36 In this reaction, we see of course carbomyl phosphate combines with ornithine to make citrulline with the release of a phosphate. 01:44 The enzyme is of course ornithine transcarbamoylase. 01:48 The enzyme is expressed only in the liver, no other place in the body. 01:52 It is the most commonly deficient enzyme that occurs in the urea cycle. 01:56 And I'll take later about how deficiencies of enzymes in the urea cycle relate to human disease. 02:02 The reason it's the most commonly deficient enzyme is that it's an X-linked inheritance. 02:06 It's found in the X chromosome which means that males are much more likely to suffer this deficiency than females. 02:13 In severe deficiency, ammonia levels can rise to very lethal levels if it's left untreated. 02:18 And the way that this is treated is commonly with liver transplant or very low protein diets to keep the ammonia levels from becoming high. 02:27 In the production of citrulline, there are other ways that this can happen. 02:32 So we saw one way that it happened earlier in the urea cycle but it can be made by another mechanism. 02:37 And that other mechanism involves the enzyme nitric oxide synthase. 02:42 This reaction is shown here and it starts actually with arginine. 02:45 It involves the reduction of arginine and the combining of four molecules of oxygen. 02:51 This s a very complicated reaction catalyzed by the enzyme nitric oxide synthase. 02:57 The product of this reaction is two citrullines, two nitric oxides, water, and NADP. 03:03 The advantage here is that cells can use this as a way of making nitric oxide which is an important molecule in signaling and can also provide an alternate means of making citrulline that can be used in the urea cycle. 03:15 This, therefore, by passes the mitochondrial part of the urea cycle if necessary and at the same time produces nitric oxide. 03:22 Arginine is a substrate for this reaction and the use of arginine can be taking away arginine from the urea cycle if that happens. 03:31 The nitric oxides system, as I said, is important for signaling and so this is an important consideration as nitric oxide is needed. 03:39 Nitric oxide helps facilitate vasodilation which is the relaxation of the smooth muscle of the blood vessels that lead to its widening. 03:46 This ultimately results to lowering of blood pressure. 03:50 Now, the citrulline gets transported to the cytoplasm if we are still in the urea cycle as I want to follow here. 03:57 The movement of citrulline to the cytoplasm requires an ornithine-citrulline translocase. 04:03 That is an enzyme that's embedded in the inner membrane of the mitochondrion. 04:08 That is called ornithine-citrulline translocase because the two molecules are swapped. 04:15 Citrulline is moved out of the mitochondrion at the same as ornithine is moved in. 04:20 And we'll see that's important at the end of the cycle as well. 04:23 The enzyme is an antiport, as I said, moving citrulline out and moving ornithine in. 04:28 The transport protein is needed for both parts of the urea cycle. 04:33 So, an absence of this from the mitochondrium has very severe consequences a person could not run the urea cycle. 04:40 A deficiency that translocase mimics many of the other enzyme deficiencies that we see in the urea cycle particularly that of a defective or deficient ornithne transcarbamoylase. 04:52 Now, this condition at birth can be more serious that its onset in adulthood. 04:57 During the development of the brain, the toxic ammonia needs to be release and needs to be removed. 05:03 And so, if the urea cycle cannot run, then there are problems. 05:06 Adult onset conditions are working with an existing brain and don't have such severe consequences.
About the Lecture
The lecture Carbamoyl Phosphate Synthetase Reaction and Citrulline by Kevin Ahern, PhD is from the course Amino Acid Metabolism.
Included Quiz Questions
Which of the following is true regarding ornithine transcarbamylase?
- It catalyzes the formation of citrulline.
- It catalyzes the formation of ornithine.
- It requires ATP.
- A deficiency has an autosomal recessive pattern of inheritance.
- It is the least commonly deficient enzyme in the urea cycle.
Which is the alternate pathway by which citrulline is formed?
- Nitric oxide synthase can be used.
- Arginine is produced.
- Nitric oxide is required.
- One citrulline is produced.
- ATP is required.
Which of the following enzymes is expressed in the liver cells only?
- Ornithine transcarbamylase
- Argininosuccinate synthetase
- Argininosuccinate lyase
- Arginase
- Carbamoyl phosphate synthetase
Citrulline gets transported out of the mitochondrion with the help of which of the following enzymes?
- Ornithine-citrulline translocase
- Ornithine translocase
- Citrulline translocase
- Argininosuccinate translocase
- Sodium/Potassium ATPase
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |