Playlist
Show Playlist
Hide Playlist
2,3 BPG
-
Slides Hemoglobin Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
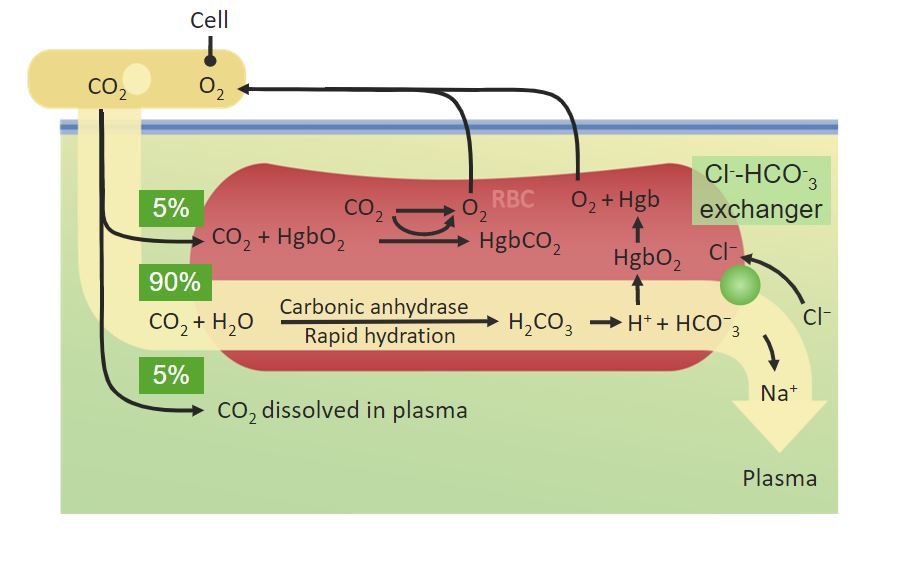
00:01 Well, it turns out that there is yet another molecule that plays an important role in the process of release of oxygen. 00:09 The molecule is what you see in the screen its 2,3 BPG or 2,3 bisphosphoglycerate. 00:15 This molecule is a byproduct of the process of glycolysis, the breakdown of sugar and it’s a relatively minor by product, it’s not made in the individual reactions of glycolysis but as a byproduct in the synthesis of one of the molecules, that’s not really important. 00:33 But what is important is that it’s made at a low level when cells are going through metabolism of glucose. 00:40 If you are exercising heavily like you might be in the race or the marathon run, then you're more likely be producing more of this 2,3 BPG than if you are sitting around eating pizza, drinking beer and watching television. 00:55 Exercising muscle tissues produce acid, they produce carbon dioxide and they produce 2,3 BPG. 01:04 Now this molecule works in a little bit different way than the other two. 01:09 It works by binding in the hole of the doughnut of hemoglobin. 01:14 So when we look at the structure of hemoglobin, the very sera where there's a cavity, there's nothing in that cavity and it turns out that’s exactly where 2,3 BPG binds. 01:25 What 2,3 BPG does is when it binds in that cavity, it converts and stabilizes hemoglobin in the T state. 01:34 Now, the T state you may remember is the state that does not like to have oxygen, so by 2,3 BPG binding into the hole of the hemoglobin, it’s favoring hemoglobin getting rid of as much oxygen as it can. 01:51 So, here’s a third signal that it’s a rapidly metabolizing cell, that the hemoglobin needs to let go of its oxygen and it does that. 02:00 If we measure the same curves that we saw before, now changing the pH, the concentration of 2,3 BPG and the concentration of carbon dioxide, the more the pH falls, the more 2,3 BPG is added and the more the carbon dioxide increases, the more the curves move to the right favoring the release of oxygen. 02:24 So, rapidly metabolizing cells produce acid. 02:31 Rapidly metabolizing cells release carbon dioxide and rapidly metabolizing cells release 2,3 BPG. 02:38 All these favor oxygen release from hemoglobin, so rapidly metabolizing cells get the oxygen they need as much as possible. 02:45 Now 2,3 BPG is interesting in another perspective. 02:49 If we think about hemoglobin bound to that 2,3 BPG with the 2,3 BPG in the hole of hemoglobin, we see hemoglobin in the T state and you might wonder what happens if hemoglobin gets back to the lungs with that 2,3 BPG stuck in the middle of it, because the 2,3 BPG favors the T state it’s less likely that the hemoglobin is going to bind to oxygen. 03:13 Well, normally in the course of things as hemoglobin is exiting the tissues that it is in, hemoglobin will let go of the 2,3 BPG. 2,3 BPG is grabbed by the cells that this hemoglobin is passing through in the blood system and the cells grab the 2,3 BPG and use it. 03:31 So the normal course of things hemoglobin gets back to the lungs and it’s lost its 2,3 BPG. 03:38 That’s not true for smokers. Smokers have a lot of 2,3 BPG in their blood, much more than nonsmokers. 03:47 So what happens is if hemoglobin lets go of its 2,3 BPG and if it’s the blood of a smoker, there's more 2,3 BPG present to go back and bind to hemoglobin, that makes it much more likely that 2,3 BPG will be bound to hemoglobin when the hemoglobin gets back to the lungs. 04:06 These locks hemoglobin in the T state when it’s in the lungs and the oxygen carrying capacity of hemoglobin in the lungs is reduced because more of it is in the T state in the blood of a smoker. 04:18 Now another side effect of that happens with smokers is it’s very important to understand with respect to hemoglobin, is that hemoglobin -- that smokers often times have a higher concentration of carbon monoxide in their blood that comes from the smoking of the cigarettes. 04:34 Carbon monoxide looks to hemoglobin an awful lot like oxygen and the reason carbon monoxide is poisonous, one of the reason it’s poisonous is because it competes with oxygen for binding to the iron on hemoglobin. 04:49 So the smokers losing on two accounts, they're losing on the carrying capacity of oxygen by hemoglobin because of the 2,3 BPG and for the hemoglobin that has no 2,3 BPG is much more likely that it will have carbon monoxide at that point.
About the Lecture
The lecture 2,3 BPG by Kevin Ahern, PhD is from the course Amino Acid Metabolism.
Included Quiz Questions
Which of the following is true regarding 2,3 bisphosphoglycerate (2,3 BPG)?
- It is produced by rapidly metabolizing tissue.
- It binds the heme portion of hemoglobin.
- It locks hemoglobin into the R state.
- It increases hemoglobin's ability to carry oxygen.
- Increases in 2,3 BPG shift the oxygen-hemoglobin dissociation curve to the left.
Which of the following is true regarding smokers?
- They are more likely to have increased 2,3 BPG levels.
- 2,3 BPG binds myoglobin.
- 2,3 BPG levels fall.
- High 2,3 BPG levels favor the R state, so oxygen is retained.
- Low levels of 2,3 BPG favor the T state, so oxygen is released.
Which of the following is true regarding carbon monoxide?
- It is higher in the blood of smokers.
- It binds to the donut of the hemoglobin.
- It binds hemoglobin in the same place as carbon dioxide
- It increases hemoglobin's oxygen-carrying capacity.
- It helps hemoglobin release oxygen to rapidly metabolizing tissues.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Great explanation about the Bohr effect, thank you very much doctor