Playlist
Show Playlist
Hide Playlist
Type II Hypersensitivity Reaction
-
Slides Immune-mediated Diseases Type II.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
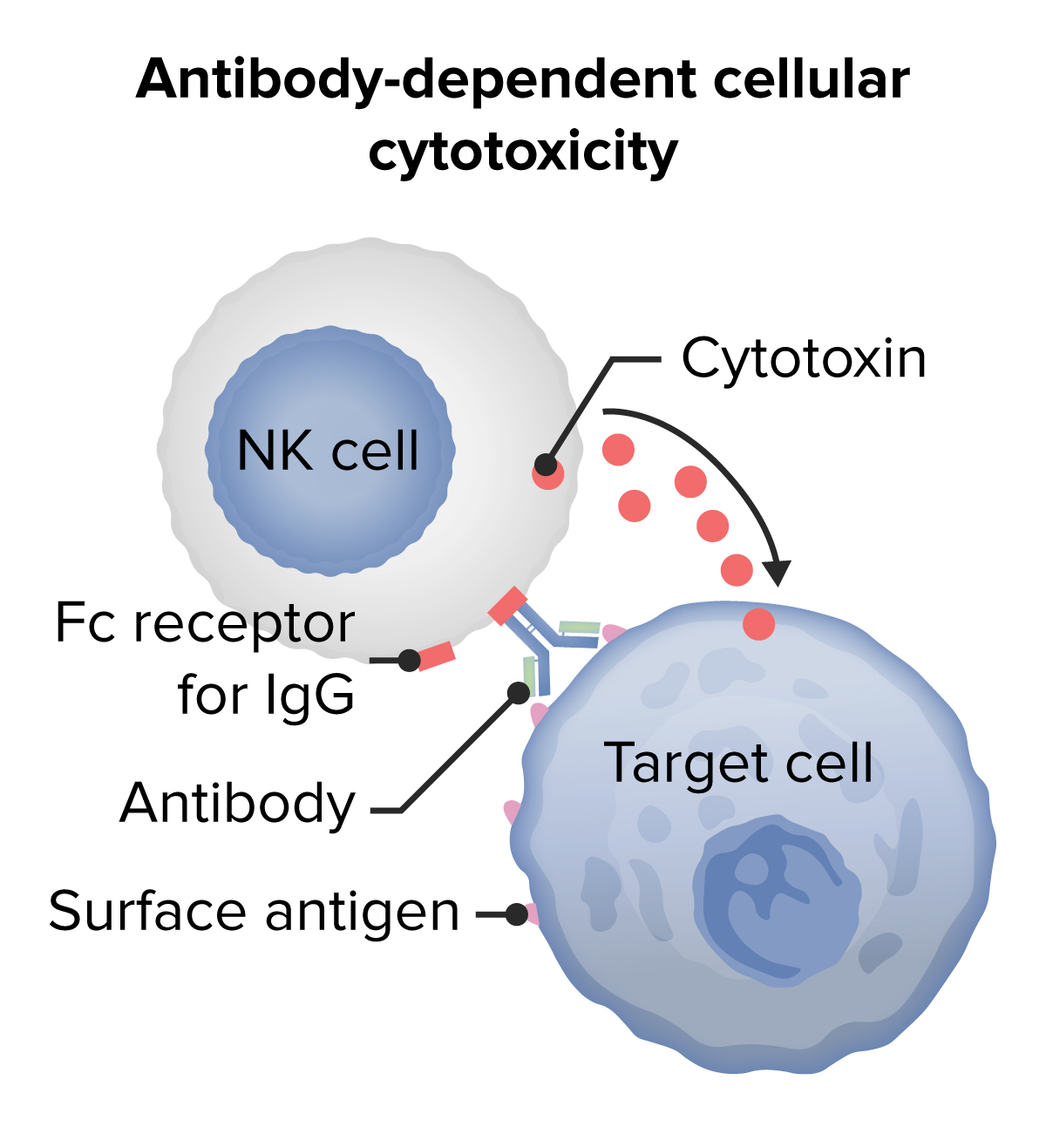
00:00 Welcome back. 00:02 Previously we talked about immediate type hypersensitivity or type 1 hypersensitivity driven by immunoglobulin E. 00:09 Now we're going to talk about type 2, which are due to immunoglobulin antibody bound to fixed surface molecules. 00:19 So type two hypersensitivity. 00:22 Let's see how this happens. 00:24 If I have a cell that I happen to have antibody bind to, that will activate complement and we will get on the surface of the cell C3b. 00:36 Remember C3b is one of those fragments that gets generated during the activation of complement. 00:42 Antibody binding either to the C3b or to the initial antigen is going to cause opsonization and lisis. 00:54 So, here we have a macrophage, that's got an Fc receptor, bound now to the antibody that is recognised a particular antigen or via the C3b receptor and that macrophage is going to ingest, degrade and kill that cell. 01:11 So, we can imagine that if this orange blob all the way on the left is a red cell, then I'm going to have the loss of those red cells by having an antibody that binds to it. 01:22 Which is exactly what we have here. 01:24 When we talk about autoimmune hemolytic anaemia. 01:28 We have a red cell, it's got a variety of surface antigens. 01:32 And if we break tolerance, or if we have antibodies to those surface molecules, we will bind the antibody. 01:40 How might that happen? Well, in fact, if we give the wrong blood group, we've give an A type blood to a B type person, they will have circulating A antibodies, which will bind to the surface of the red cell. 01:55 That binding of the antibody will cause complement activation and deposition with rupture the cells. 02:01 So that's the autoimmune hemolytic anaemia, but we've also made these cells tasty. 02:06 We've opsonized them by binding complements C3b and by having an antibody present with a rearranged Fc portion, and it will be pulled out. 02:14 Those cells will be pulled out by macrophages as they go through the liver and the spleen. 02:20 So that's what happens if we have antibodies against red cells. 02:24 And it can be a transfusion reaction. 02:27 It can be due to mom having antibodies against baby's Rh. 02:32 Or it could be a primary autoimmune disorder, autoimmune hemolytic anaemia. 02:38 What does this look like? Not only is the patient anaemic, we're destroying red cells, were eating up red cells, were fragmenting red cells. 02:47 So there's lower numbers of red cells, but we're also getting fragments. 02:50 And the arrow that's pointing to that funny looking red cell at the top is actually identifying a cell that was coated with antibody and C3b. 03:01 And as it went through the gauntlet of macrophages in the red pulp in the spleen, bits and pieces of that red cell got nibbled away at. 03:10 And so we have, in fact, just the sides. 03:12 We've got fragmented red cells, and that's just an example on our peripheral smear. 03:18 Okay, that's on circulating cells. 03:20 That one's pretty easy to wrap our minds around. 03:22 But what about antigen that's on a tissue, such as a basement membrane. 03:28 So here we have a basement membrane that's indicated along the bottom and we have particular proteins that are inserted into it. 03:34 And oh my goodness, now we have antibodies that are bound to those. 03:37 Those antibodies will have rearrangement of the Fc portion and now neutrophils or macrophages, things that have Fc receptors will be able to bind. 03:48 We will also by virtue of having that antibody bound activate complement. 03:52 So we're going to get complement fragments that will settle down onto the surface including C3b and bind to those proteins, while also make complement byproduct. 04:02 So C3a and C5a that will activate mast cells and recruit neutrophils and activate inflammatory response. 04:09 So we'll get neutrophils that come in response to the complement. 04:13 But we will also have the cells trying to ingest what is functionally a very large surface and they won't be able to do so. 04:21 So they'll be dumping their mediators, their proteases, their arachidonic acid metabolites directly onto the tissue, potentially causing damage. 04:31 Remember, our old friend the natural killer cell, has on its surface Fc receptors that can bind to bound antibody on the surface of a target. 04:41 So the NK cell, as you see there, is interacting with the orange blob in the upper right hand corner via antibody receptor interaction, so called antibody dependent cell mediated cytotoxicity. 04:52 So binding antibody to tissues will cause cell death in that way as well. 04:58 And this notion of frustrated phagocytosis is pretty important to understand. 05:03 So here, this is a normal neutrophil with something it can eat. 05:08 It can get its pseudopods around. 05:10 So it's recognised that microbe via antibody bound to it, or via complement receptor fragments. 05:19 And it now will ingest phagocytosis and it will be able to get it completely enclosed. 05:26 And now, it can fuse lysosomes with that phagosomes and destroy whatever it ate. 05:33 That's great for smaller things but if we an antibody binding to big things, like a basement membrane, neutrophil comes up, and wants to do same thing. 05:41 It's binding through its Fc receptor and its complement receptor and it's ready to rock and roll except that it can't ever get it all inside. 05:50 So now what it does is it basically spews its lysosomal content on to the surface. 05:57 It's frustrated phagocytosis, it can't get its arms around it. 06:01 And now I will get damaged because of the release of extracellular enzymes and other mediators on to the tissue. 06:09 Oh my god, does this happen? Oh yes, it does.
About the Lecture
The lecture Type II Hypersensitivity Reaction by Richard Mitchell, MD, PhD is from the course Immune-mediated Diseases.
Included Quiz Questions
Which of the following mechanisms is not involved in the pathogenesis of type II hypersensitivity reactions?
- T-helper-1 cell-mediated cytokine release
- Opsonization
- Complement activation
- Neutrophil and macrophage recruitment
- NK cell-mediated lysis
Which of the following mechanisms is involved in the pathogenesis of type II hypersensitivity reactions?
- Antibody-dependent cellular cytotoxicity
- Deposition of immune complexes in tissues and small blood vessels
- Release of vasoactive amines and cytokines from mast cells
- Cell-mediated cytotoxicity
- Autophagy
Which of the following diseases is an example of a type II hypersensitivity reaction?
- Autoimmune hemolytic anemia
- Polyarteritis nodosa
- Asthma
- Contact dermatitis
- Rheumatoid arthritis
Which of the following complement subtypes is expressed on the surface of the target cells during a type II hypersensitivity reaction?
- C3b
- C5b
- C3a
- C6
- C7
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |