Playlist
Show Playlist
Hide Playlist
Proximal Tubule: Ion Transport
-
Slides 03 Reabsorption1 UrinarySystem.pdf
-
Download Lecture Overview
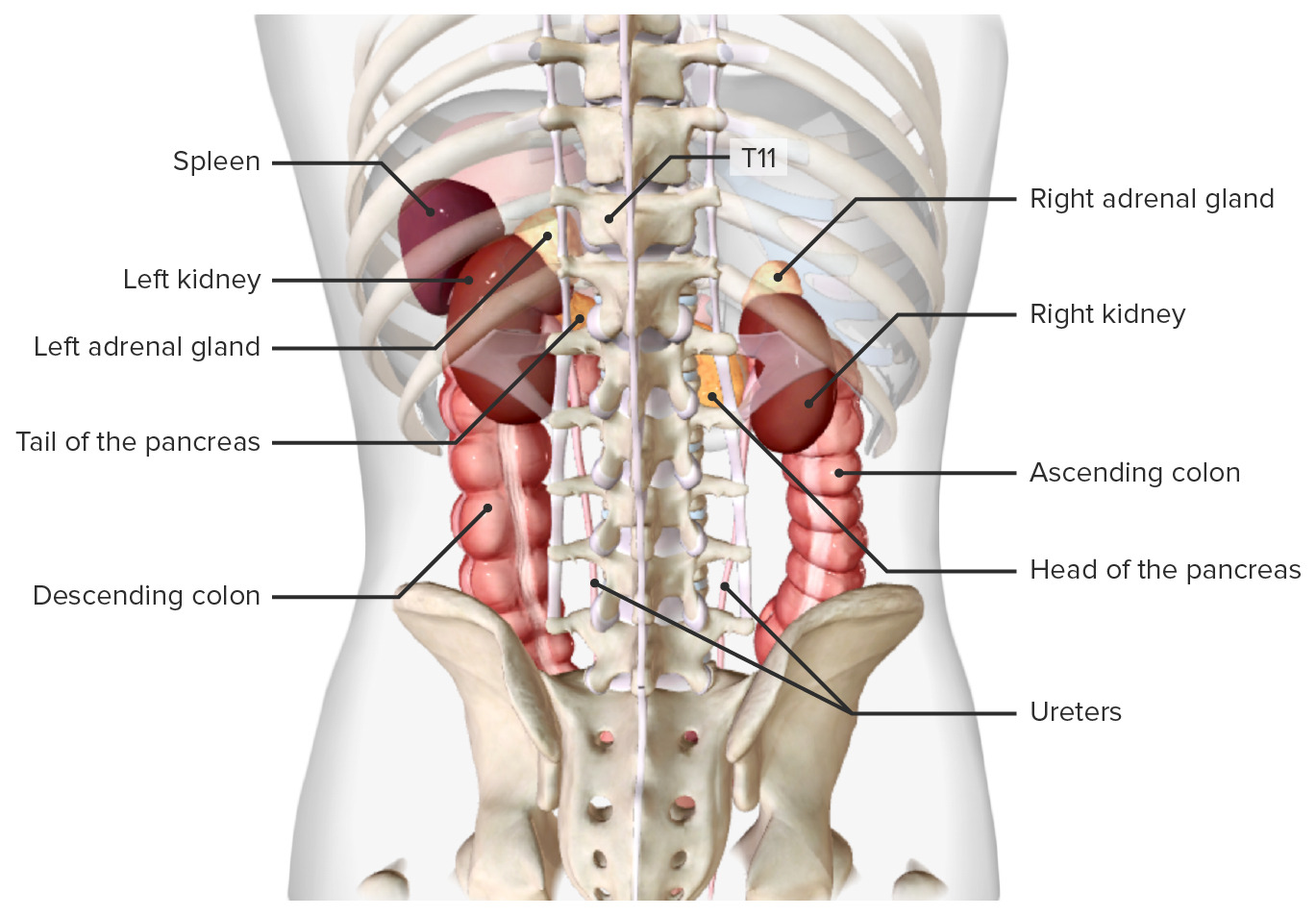
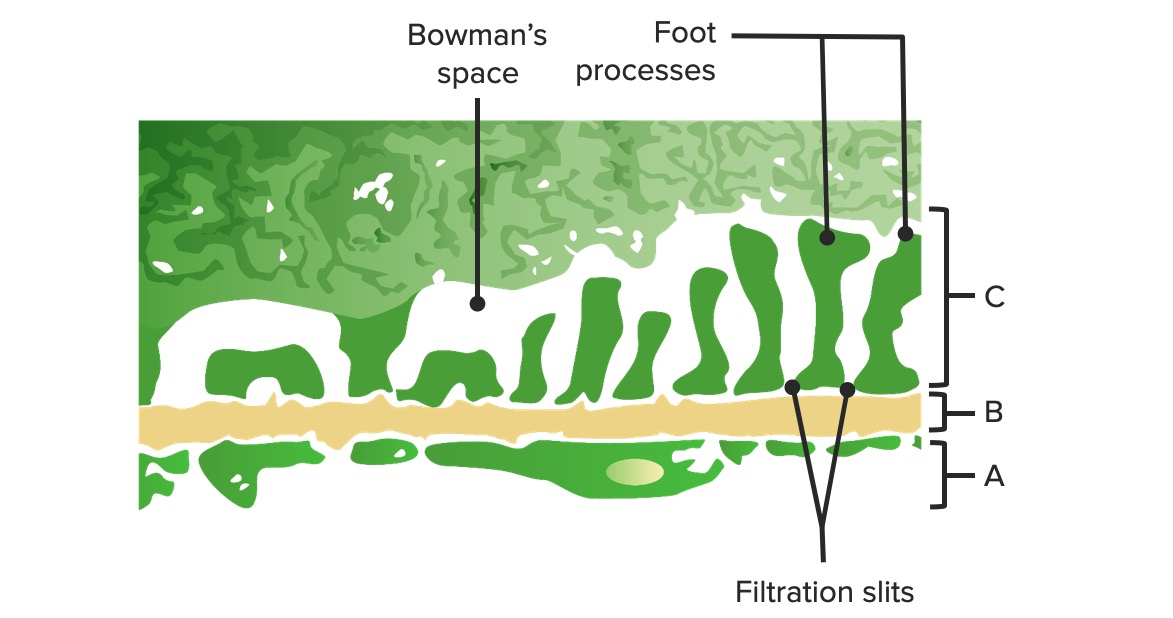
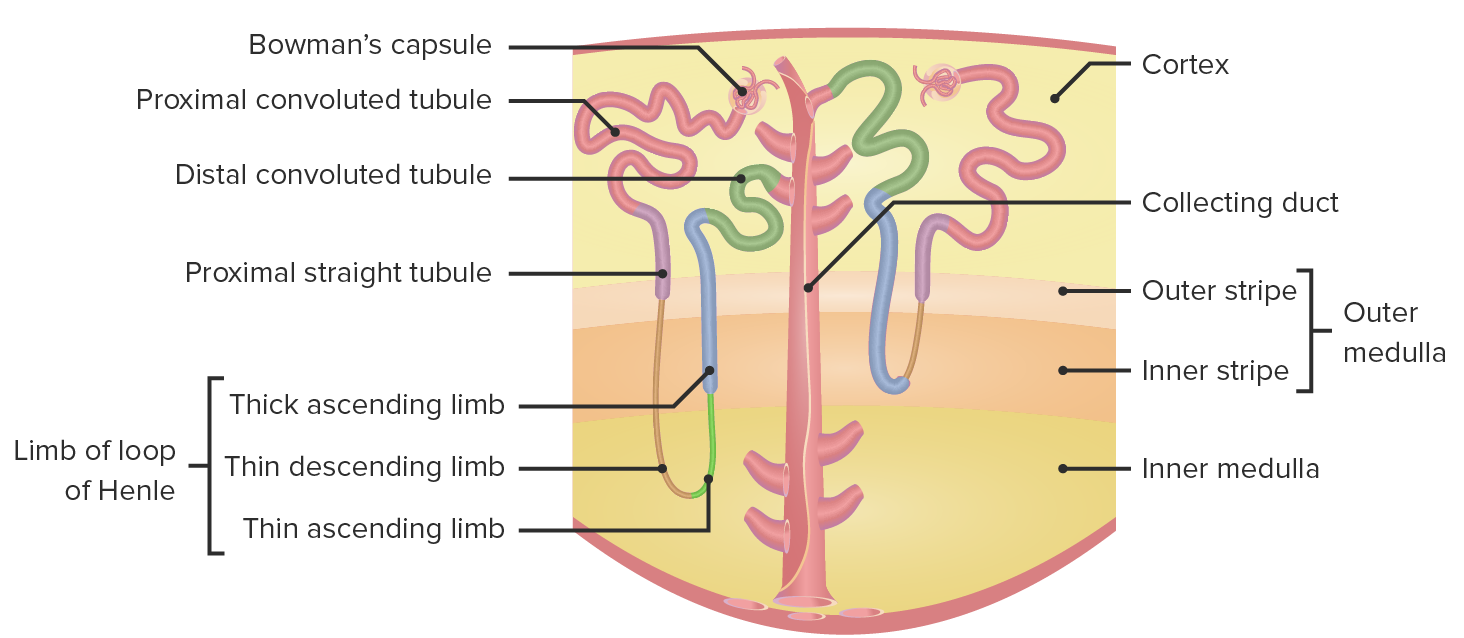
00:01 Now, let’s deal with some of the ions individually in terms of their transport across the proximal tubule. 00:09 Sodium is one of the biggest ones to discuss because it serves as a driving force for many of the other solutes that cross the apical membrane. 00:19 What starts this? This is governed by this sodium-potassium ATPase. 00:25 So as sodium starts to come down the renal tubule, there’s a concentration gradient that’s set-up across the renal tubule cell. 00:36 That concentration gradient will be then linked with other solute molecules to allow them to be co-transported. 00:46 And then as they cross the basolateral membrane, that’s usually done by the sodium-potassium ATPase and/or other transport mechanisms. 00:55 So many other molecules will use this sodium driving force to allow for its transport, and that’s all through a co-transport mechanism. 01:08 The interesting thing about sodium that you always need to keep in mind is, as sodium moves, so does water want to. 01:17 So if you have a leaky epithelial membrane, your water will always follow the sodium transport – and in this case, sodium reabsorption. 01:27 Other ions such as chloride – these follow again a gradient across the apical membrane. 01:35 But interestingly, they use a paracellular flow mechanism rather than a lot of transporters. 01:45 This is set up by this transepithelial voltage gradient. 01:52 Since chloride is a negatively charged molecule, it will want to travel towards areas that are positively charged. 01:58 Remember, opposites attracts in this case. 02:02 The negatively charged chloride would want to travel over to the positively charged areas. 02:10 This will occur paracellularly or between the cells. 02:16 There are other molecules, however, that may have exited the basolateral membrane, and these will want to travel in the direction of their electrocurrent gradient. 02:26 So if it’s a positively charged molecule, it may want to leak back into the epithelial tubule. 02:33 Calcium and magnesium – these are brought into the blood in the proximal tubule. 02:40 This is done via these transepithelial voltage differences, as well as that solvent drag. 02:47 Remember, solvent drag was the process by which water moving paracellularly through this – between the 2 cells – also drag along or pulled along with it some other molecules. 02:59 You can think of this a lot like a flood. 03:02 So after a flood, maybe a tree falls over, and then the tree gets dragged down the river along with it. 03:08 that’s the same thing that happens with solvent drag. 03:11 As the water flows across these paracellular regions, they drag some things with them. 03:17 A lot of this calcium is reabsorbed in this manner – about 65 percent. 03:22 A little bit less for magnesium, which is only about 15 percent of its filtered load is reabsorbed in the proximal tubule. 03:30 So we’ll have to revisit both of these 2 ions later on in the nephron to see how they’re reabsorbed. 03:40 Potassium transport is one that is a little bit complex. 03:45 It’s going to utilize 2 basic mechanisms that are very similar to the other divalent cations that we just talked about with calcium and magnesium. 03:54 They use a solvent drag component or this transepithelial voltage difference. 04:01 A lot of the potassium is brought across in this manner – around 80 percent. 04:08 So that’s a lot more than the calcium, which was 65, and the magnesium, which was 15. 04:14 So a greater degree of the total filtered load is transported across by these 2 mechanisms. 04:22 Solvent drag being the most important of these, and that is as the water travels through these – between the cells – they will pull along with it potassium. 04:34 Later in the proximal tubule section, there is a greater transepithelial gradient. 04:41 And remember, we have a positive charge and a negative charge. 04:45 Early on, chloride moved from a negatively charged molecule over to a positive charge. 04:52 As we move down the proximal tubule to more of the proximal straight component, the gradient switches and you have a negative charge on the basolateral side and a positive charge on the apical. 05:04 This very small transepithelial gradient causes a little bit of potassium to move across. 05:11 So what do you do with all of these solutes that have been moved across the basolateral membrane? Whether it was done through a transcellular or paracellular mechanism, you have all of these solutes that are hanging out in the interstitial fluid. 05:25 Well, you have to get them into the blood, and you have to have the blood take them away. 05:30 And that is what’s going to be involved for this particular process – you’re picking up the solutes and you’re bussing them away. 05:40 How does this process work? Well, if you think about the starling forces that we’ve revisited a couple of times during this course – if you think about starling forces in a normal systemic capillary, you usually have an area that involves some filtration, and then an area that involves reabsorption. 06:00 So in this case, the filtration is pushing fluid out of the capillary, and the reabsorption is bringing it – sucking it back in. 06:10 Unlike the systemic capillaries, peritubular capillaries are going to involve reabsorption. 06:17 They reabsorb throughout the length of the capillary. 06:22 So this process occurs both on the arterial side of the capillary and on the venous side of the capillary bed. 06:30 It's all about reabsorption. 06:32 So this is the area in which all of the solutes that moved across the basolateral membrane, all the solutes that traveled via solvent drag, ended up in the interstitial space. 06:44 What you’re doing is basically capturing all of them, moving them into the peritubular capillaries, and they’re going to be moved out into the blood.
About the Lecture
The lecture Proximal Tubule: Ion Transport by Thad Wilson, PhD is from the course Urinary Tract Physiology.
Included Quiz Questions
Which of the following mechanisms are used to reabsorb potassium along the proximal tubule?
- Solvent drag and simple diffusion
- Apical potassium channels
- Basolateral sodium-potassium ATPase
- Basolateral potassium channels
Which of the following is the driving ion for the transcellular flow of molecules across the apical membrane of cells in the proximal convoluted tubule?
- Sodium
- Glucose
- Calcium
- Potassium
- Phosphate
Which molecule is transported via a paracellular mechanism driven by the electric gradient created by sodium reabsorption?
- Chloride
- Potassium
- Calcium
- Magnesium
- Phosphate
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |