Playlist
Show Playlist
Hide Playlist
Pediatric Thalassemia
-
Slides Thalassemia Pediatrics.pdf
-
Download Lecture Overview
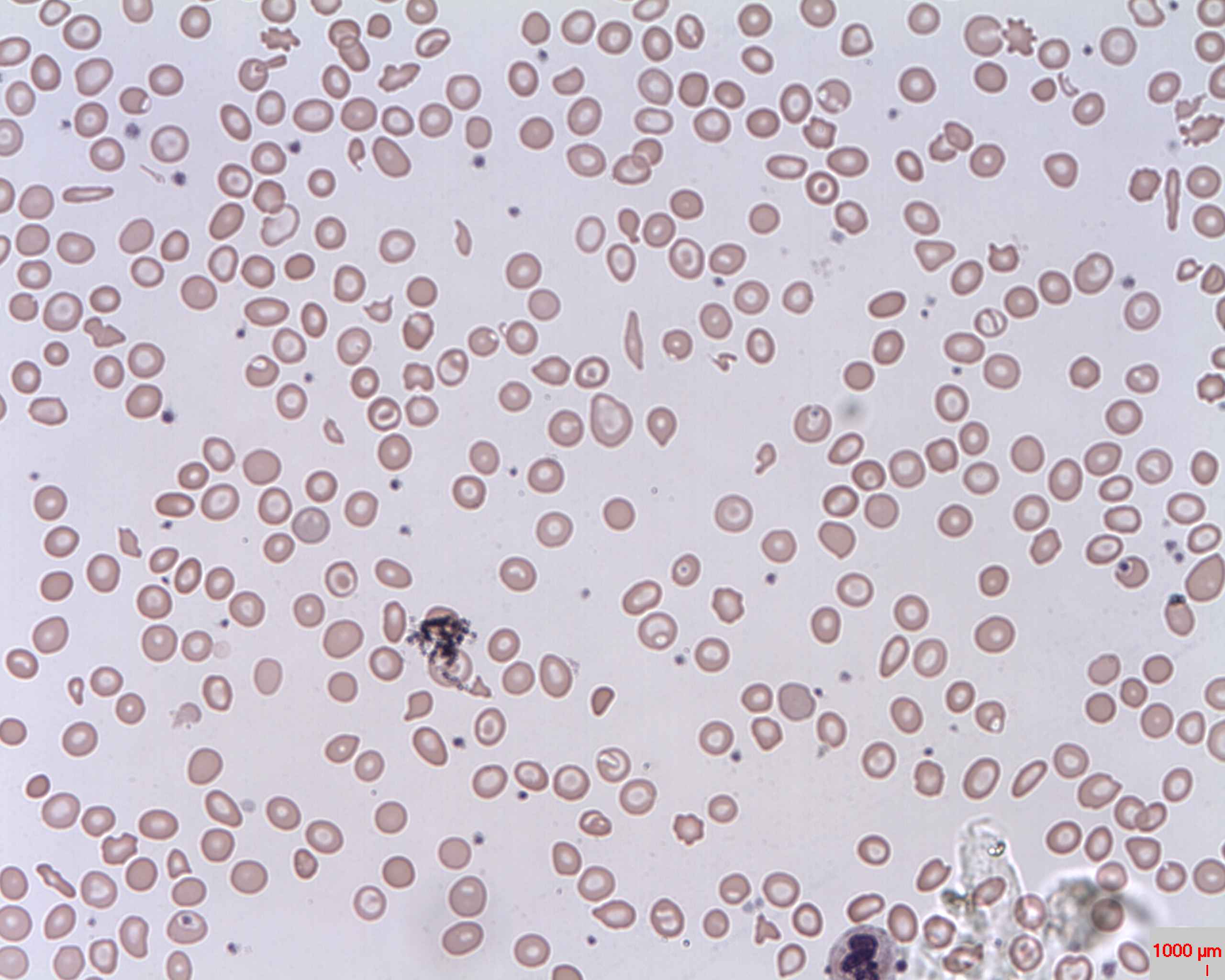
00:01 In this lecture, we’re going to discuss thalassemia in children. 00:06 So thalassemia is an inherited microcytic anemia. 00:11 It happens as a result of an abnormality in hemoglobin synthesis in either the alpha or the beta subunit. 00:19 And mutations in the alpha or beta subunit are usually autosomal recessive. 00:25 So patients can be carriers of disease. 00:28 Let’s remember some basic science about the hemoglobin molecule. 00:32 Each hemoglobin molecule has two alpha and two beta subunits. 00:38 But in the genome, there are two copies of the beta gene, these are on chromosome 11 and four copies of the alpha gene, these are found on chromosome 16. 00:50 So when the patient has an abnormal mutation in one of these subunits, it leads to an excess of the unmutated subunit. 01:01 That buildup of unaffected subunits results in cells breaking open prematurely and hemolysis. 01:11 So we tend to see alpha thalassemia in areas where there is more malaria or among people who historically were more likely to get malaria, because having alpha thalassemia, much like having sickle cell trait, was somewhat protective of the disease. 01:31 The severity of alpha thalassemia depends on how many of those four alleles that are in the genome are affected. 01:39 Let’s go through that. 01:41 If a patient has only 1 alpha affected, they have dash and then a normal alpha and then two other normal alphas on the other gene, this patient is found to have alpha thalassemia minima. 01:57 These patients are essentially silent gene carriers. 02:00 They don’t have any symptoms. 02:02 If on the other hand, they have alpha/alpha and on the other chromosome, two mutations, this is alpha thalassemia minor. 02:10 This is more common in Asian populations especially Southeast Asia and these patients may have a mild microcytic anemia but are generally functioning normally. 02:21 Alternatively, they could have 1 mutation on each chromosome, this is also alpha thalassemia minor, but we see this variant more in Africans, again, a mild microcytic anemia. 02:32 If they have 3 mutations, this is called hemoglobin H disease. 02:37 These patients will have a significant microcytic anemia and generally hepatosplenomegaly. 02:43 Alternatively, if they have mutations in all four alpha genes, this is alpha thalassemia major. 02:51 These patients will be edematous as a newborn and often die before birth. 02:56 They may have hydrops fetalis. 03:01 So let’s look at beta thalassemia now. 03:04 Remember, beta thalassemia, there is only two alleles, but there are two types of mutations that you can have to a beta subunit. 03:12 You can have beta plus where there is less production of that protein from the gene or you may have beta zero, in which there is no production of the protein at all from this gene. 03:25 So we now have different variations of disease. 03:29 If you are beta plus/beta or beta zero/beta, this is called beta thalassemia minor. 03:36 This presents usually as a microcytic anemia. 03:40 Alternatively, if you are beta zero/beta plus or beta plus/beta plus, this is beta thalassemia intermedia. 03:48 These patients have a microcytic anemia and occasionally require transfusions. 03:53 More severely, if you have two beta zero copies, this is really bad. 03:57 This is called beta thalassemia major. 04:00 These patients have severe anemia. 04:03 They usually have hepatosplenomegaly. 04:05 They have significant iron overload. 04:08 They have bony deformities -- We’ll go through that disease in more detail. 04:12 So let’s look at the basic clinical presentation and pathophysiology of beta thalassemia. 04:19 In beta thalassemia, because they have mutated betas, they have an accrual of insoluble alpha globin. 04:26 That alpha globin forms inside that cell and that causes an abnormality in the erythroblast, so that there is ineffective hematopoiesis and the bones are incapable of making adequate red blood cells. 04:38 And most of those erythroblasts will die inside the bone marrow. 04:42 Among those that escape, there will be increased extravascular hemolysis specifically in the spleen. 04:49 And these cells will collect there and patients may get splenomegaly. 04:54 So let’s look a little bit deeper, however. 04:58 In patients where there is ineffective erythropoiesis and most of those erythroblasts are dying in the spleen, the patient is still getting a normal amount of dietary iron. 05:08 They will have increased iron absorption and they may develop iron overload. 05:14 These patients will also have extravascular hemolysis which will result in anemia. 05:20 Remember, because these patients are having anemia, they may require blood transfusions and blood transfusions obviously also contain iron, further contributing to the systemic iron overload. 05:34 Furthermore, the tissue hypoxia results in an upregulation of erythropoietin release from the kidney and EPO that’s released will encourage not only bone marrow expression, but it will encourage that bone marrow expression in such a way that there will sometimes be skeletal deformities as a result of that marrow constantly being encouraged to undergo erythropoiesis. 06:01 This can lead to a rather startling finding. 06:05 Specifically, patients may develop bone marrow that is so aggressive that you can see these deformities not only grossly when you approach the patient, but on x-ray. 06:16 This is a classic skull x-ray of an infant with beta thalassemia major, beta zero/beta zero. 06:23 You can see this picture has something called a hair-on-end appearance and that skull is undergoing hematopoiesis. 06:31 Additionally, this can result in abnormal facies and you can see a small appearing nose, very prominent maxillae as the areas of that skull that are requiring to do hematopoiesis have expanded. 06:46 So the diagnosis of thalassemia is basically made by hemoglobin electrophoresis and that’s how we can decide what type of thalassemia that patient has. 06:57 The treatment is fairly simple. 07:01 For patients with alpha thal minima and minor and for patients with beta thal minor, we typically give folate. 07:09 This is because they have dietary increase need for folate because of hematopoiesis. 07:14 For patient with more severe disease such as hemoglobin H disease or beta thalassemia intermedia, we will not only do folate but patients will require periodic transfusions. 07:25 If a patient has yet more severe disease such as beta thalassemia major, these patients may require transfusions as much as every 2-3 weeks. 07:36 As a result, they require deferoxamine to chelate the excess iron, so it doesn’t buildup in their tissues. 07:44 In severe cases, we may move on to bone marrow transplant. 07:49 In alpha thalassemia major, these patients are very sick. 07:54 They require in utero transfusions and eventually a bone marrow transplant. 08:00 So that’s my review of thalassemias in children. 08:05 Thanks for your attention.
About the Lecture
The lecture Pediatric Thalassemia by Brian Alverson, MD is from the course Pediatric Hematology. It contains the following chapters:
- Thalassemia
- α Thalassemia
- β Thalassemia
- Diagnosis and Treatment
Included Quiz Questions
Which of the following leads to hemoglobin H disease?
- Inheritance of 3 mutated alpha subunits of hemoglobin
- Mutations in 2 alpha subunits of hemoglobin on the same chromosome
- Mutations in a beta and an alpha hemoglobin subunit
- Both alpha thalassemia and sickle cell disease
- Both beta-thalassemia and sickle cell disease
Which type of anemia is most likely in patients with thalassemia?
- Microcytic hypochromic
- Normocytic normochromic
- Normocytic hypochromic
- Microcytic hyperchromic
- Macrocytic hypochromic
Which of the following results from inheritance of 2 mutated hemoglobin alpha chain genes?
- Alpha thalassemia minor
- Silent disease
- Hemoglobin H disease
- Alpha thalessemia major
- Alpha thalassemia minima
Which of the following thalassemias results from beta0/beta+ inheritance?
- Beta thalassemia intermedia
- Beta thalassemia minor
- Beta thalassemia major
- Alpha thalassemia minor
- Hemoglobin H disease
Which of the following is the major cause of the skeletal deformities in patients with beta thalassemia major?
- Bone marrow overstimulation
- Iron overload
- Multiple blood transfusions
- Bone marrow suppression by viral infections
- Secondary hemochromatosis
Which of the following is most appropriate initial diagnostic test in an anemic child suspected of having thalassemia?
- Serum hemoglobin electrophoresis
- Bone marrow aspiration
- X-ray of the long bones
- Serum protein electrophoresis
- DNA analysis
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Excellent overview of the disease, the genotypes and the treatments.