Playlist
Show Playlist
Hide Playlist
Myocardial Infarct: Timing and Biomarkers
-
Slides Complications of Atherosclerosis.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
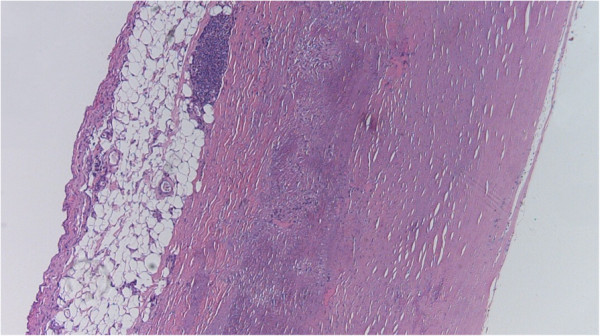
00:01 So let's talk about the timing of events that occur after loss of perfusion, specifically to the heart. 00:07 And we're going to emphasize the heart because, in fact, cardiovascular death is going to be one of the major forms of ischemic infarction injury across the human population. 00:20 So within seconds, after we cut off the blood supply, say with a thrombus or a plaque rupture in a coronary artery, within seconds, the tissues, the myocardium, that is being perfused, by that blood supply turns over into anaerobic metabolism. 00:41 So we are now not, no longer using oxygen to generate ATP. 00:48 Within a couple minutes, even that is not able to maintain ATP generation, at a level sufficient to maintain contractility. 00:58 So within a couple minutes, we have lost the ability of the heart to contract. 01:02 It's not dead, but it's not functional. 01:06 And therefore, we will be having problems with regard to generating enough blood supply to go to the brain or whatever. 01:14 The ATP is reduced to 50% of normal within 10 minutes. 01:19 So all the things we've been doing before the anaerobic metabolism, cutting down the amount of contraction, everything that we're doing to maintain ATP at some level, significantly reduced, is significant reduced within 10 to 40 minutes. 01:35 Once we get to less than about 10 percent, that's when the tissues will die. 01:41 So we will have irreversible cell injury, death, infarction beginning at 20 to 40 minutes. 01:49 Now, interestingly, if you think about the heart, it's not just cardiomyocytes. 01:53 There are fibroblasts, which are relatively resistant to ischemic injury. 01:58 But there's also vessels, endothelial cells. 02:02 And beginning to about an hour, they will also begin dying. 02:07 That means if we restore blood supply, after about an hour of ischemia, or when we are having infarction, death of the endothelial cells, then if we restore blood supply by giving a clot buster, tissue plasminogen activator, we will in fact, get bleeding into the heart. 02:26 Okay, so let's look at this, let's look at what looks like. 02:31 We have a heart, we have a thrombus, we have a kind of a brownish area where we're not getting enough blood supply. 02:39 What is going on at the vascular level is what is shown most commonly. 02:43 On the left hand side, we've seen this image in a prior session. 02:47 We have an atherosclerotic plaque, that was probably not flow limiting. 02:52 It was somewhere less than a 70% chronic stenosis. 02:56 And at some unpredictable point, that plaque ruptured. 03:01 The thin fibers cap over the surface of the atheromatous core broke. 03:06 And then we expose the underlying thrombogenic materials to all the blood, and we got a clot. 03:12 That thrombus limits blood supply downstream. 03:15 The consequence is shown on the right hand side, and the arrow is pointing to an area of infarct, a territorial area of infarct, where we've actually even had some reperfusion in an attempt to save this patient's life, and that's why we have the hemorrhage. 03:32 So that territory with the arrow is a rather large area of infarct, and is going to lead to probably a combination of things, including arrythmias but also diminished cardiac output. 03:48 The rest of the heart is not going to be able to maintain sufficient squeeze to provide blood supply to the rest of the body or to the brain. 03:55 And as a consequence, the patient has expired. 03:59 How do we measure, how do we identify this as it's happening? That's where we bring in biomarkers. 04:06 So there are a number of components within cardiac myocytes that we can measure in the circulating blood that will give us an idea whether or not we have irreversible damage. 04:18 So these are the biomarkers of acute infarction. 04:21 So on the left hand side, we have onset of myocardial infarction, that tissue is dying. 04:27 At the periphery of that zone, there are still blood vessels that are able to exchange at the periphery, things that are in the myocardium with oxygen and other stuff, As the muscles are dying in that peripheral area, the various components of the cardiac myocytes, as demonstrated here, some of the proteins - the myosins the actins, the creatine kinase MB fraction, the CKMB, troponins which are part of the actin-myosin complex and allow the filaments to slide over one another. 05:04 All of these are released into the blood supply. 05:09 And we can by drawing blood out of an arm vein, see how much damage has been done. 05:15 Clearly, if we reperfused this area, now we have lots of blood supply coming in, and we will get a big surge in these markers. 05:24 The markers are not necessarily created equal. 05:28 So we think cardiac myocytes, there is myoglobin. 05:32 Myoglobin is one of the very first proteins that we can use to measure an incipient or ongoing myocardial infarct, but it comes up and it goes down very, very quickly. 05:43 So it's not really a very good marker. 05:45 Most patients don't appear at time zero when they're having an infarct, and we can watch them and see at peak at about five hours. 05:52 The other problem about myoglobin is it's found in skeletal muscle. 05:55 It's found in a variety of tissues so it's a relatively nonspecific marker. 06:00 The next marker that has been used very commonly traditionally for many, many decades, is the creatine kinase MB fraction. 06:08 Creatine kinase, important protein for driving and generating ATP. 06:16 That creatine kinase is also found in other tissues, but there are different isoforms. 06:23 So there's an MM isoform that's found in skeletal muscle. 06:27 There's a BB isoform, that's found in brain. 06:31 The MB fraction or the MB isoform, is found predominantly, but not exclusively in cardiac muscle. 06:37 And again, this one has been used traditionally, to be a good marker for a specific injury to the cardiac myocytes. 06:46 The one now that we use most commonly in hospitals, clinics around the world is troponin and there are various subforms, isoforms of troponin that we use, but most of the ones, all the ones that we use, are specific to myocardium. 07:01 They are so much better biomarkers, they last much longer. 07:07 And we will traditionally measure, a patient comes in with chest pain. 07:11 We're not sure whether or not they're having a heart attack, a myocardial infarction. 07:16 We will measure over the next 24 to 48 hours, 3 or so troponins, each at about 12-hour intervals, identifying one - if there is an infarct, two - how much of an infarct there is, the more troponin you have released, the greater the size of the infarct and making sure that that infarct is a one time only, and that the troponin trends down at the end. 07:42 So this is what you will use on a regular basis. 07:46 And here's the reason that you use it and why we use troponins much more commonly now than CKMB and why we never use myoglobin.
About the Lecture
The lecture Myocardial Infarct: Timing and Biomarkers by Richard Mitchell, MD, PhD is from the course Atherosclerosis.
Included Quiz Questions
What is the first biomarker to appear at the onset of acute myocardial infarction (MI)?
- Myoglobin
- Troponin
- Actin
- CK-MB
- CK-BB
Creatinine kinase is an enzyme that generates the production of...
- ...ATP.
- ...CMP.
- ...cCMP.
- ...G6P.
What is a reliable biomarker for acute myocardial infarction?
- CK-MB
- CK-MM
- CK-BB
- BNP
- Myoglobin
Troponin levels increase with...
- ...the size of the infarct.
- ...the location of the infarct.
- ...the type of infarct.
- ...the composition of the infarct.
- ...the age of the infarct.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |