Playlist
Show Playlist
Hide Playlist
Movement of Larger Molecules Across Membranes: Pinocytosis
-
Slides Cellular Pathology - Movement across Membranes.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
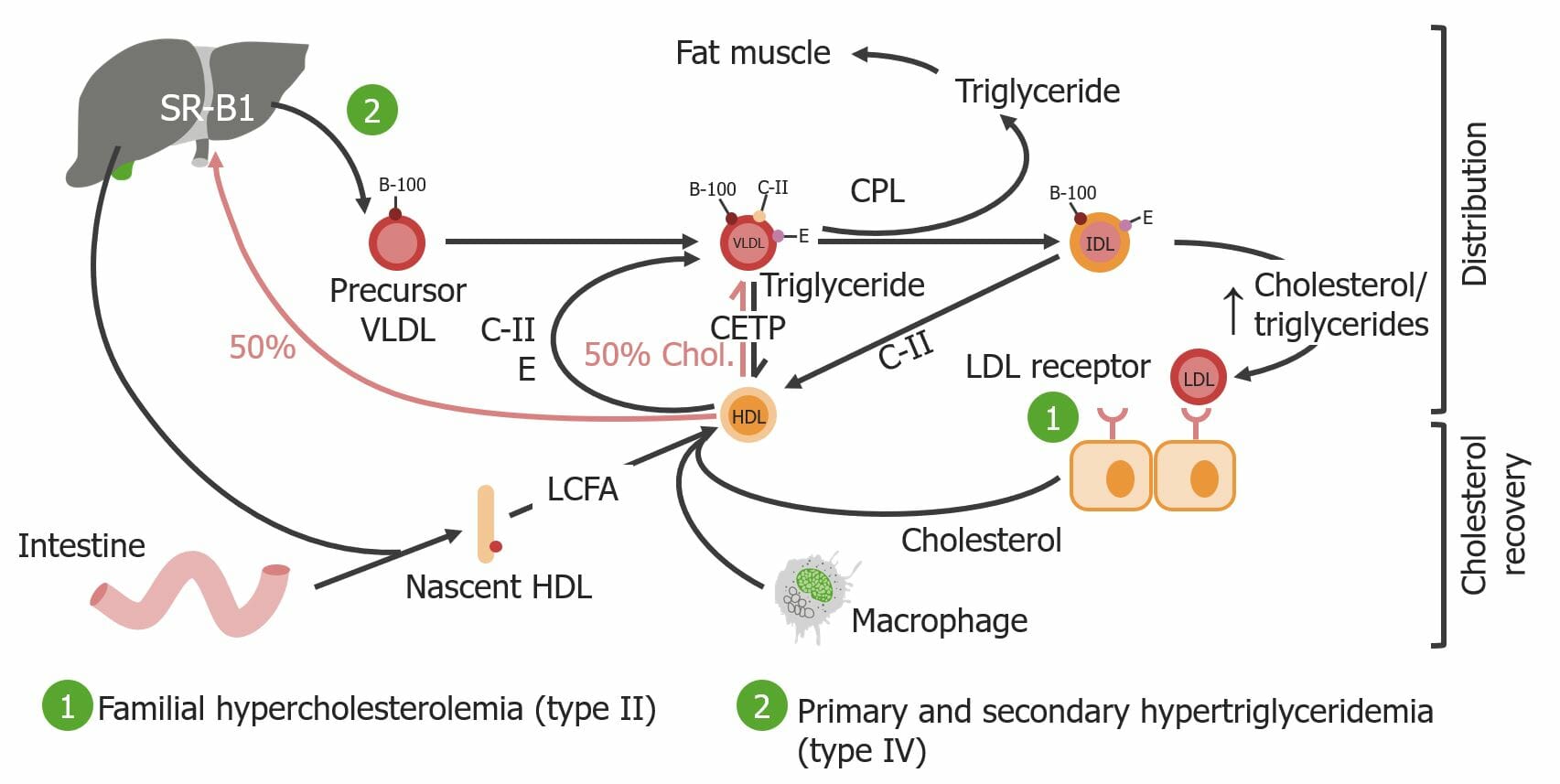
00:01 Let's talk about pinocytosis. So, literally, cell drinking. 00:06 I'm gonna use the example of the LDL receptor or the low-density lipoprotein receptor, but there are many other receptors that use this pathway, endocytosis, pinocytosis, to get their cargo into the cell. 00:22 Example -- another example is the transferrin receptor. 00:25 Transferrin is gonna be the way that we bring in iron to a cell and it binds to a specific receptor also in a receptor-mediated endocytosis pathway. 00:37 So, there are other molecules that follow this. 00:39 But LDL is going to be important because defects in this receptor or this pathway can give rise to a disease that we'll talk about in a minute. 00:48 So, we have LDL floating around that binds to its receptor. 00:55 That receptor then, once it's loaded with ligand, will go to an area of the membrane where clathrin, another molecule, somewhat similar to caveolin, but different, will bind to the vesicle that's being formed, and we get a clathrin-coated pit. 01:13 And you see it there, LDL receptor with its cargo. 01:17 That will internalize into a clathrin-coated vesicle, so it pinches off, and the clathrin is what drives that process. 01:25 And in there, we now have the receptor, and many other receptors, and their cargo. We uncoat. 01:33 So, we release the clathrin and it recycles, and we will use it for another round of another clathrin-coated pit endocytosis. 01:41 But now, that clathrin-coated vesicle will mature. 01:45 In some cases, it directly matures to become an early endosome. 01:48 In other cases, it will fuse with a pre-form structure that looks like an early endosome. 01:54 In this compartment, now we have delivered the receptor and its ligand. 01:59 That receptor, ligand, is separated one from another by virtue of pumping in protons. 02:05 The change in the pH gives dissociation. 02:08 And now, we're able to separate receptor from cargo. 02:12 In this case, free LDL. The receptor, we recycle, and will go through another round. Perfect. 02:18 What about the LDL that we took up? Well, that LDL will be targeted to lysosomes where it gets degraded into its constituent components, including the amino acids and cholesterol. 02:29 And importantly, the free cholesterol that gets generated in this process will feedback inhibit to the nucleus and say, "Quit making cholesterol receptors. 02:38 Don't make any more LDL receptors. 02:41 And also, don't synthesize cholesterol." That feedback is really important because if we don't have this process, we don't have that feedback, and we have very high levels of cholesterol. 02:54 So, let's look at a disease as promised. 02:57 Familial hypercholesterolemia. 02:59 And in this disease, there are a variety of mutations that basically either make a mutant form of the LDL receptor that doesn't bind LDL or a mutant form that doesn't internalize or a mutant form that doesn't target appropriately as it goes along the way. In that case, we do not appropriately take up LDL. 03:19 We leave it out in the extracellular space. 03:23 So, we're not getting rid of all the cholesterol thats circulating. 03:26 And not only that, but we're not giving that signal to the nucleus to quit making the LDL receptor. Boo, not good. 03:34 And we are not giving the signal that says, "Don't make any more cholesterol." So, we are now making more cholesterol. This happens in hepatocytes. 03:45 We're making lots and lots of cholesterol and we're not clearing the LDL that's out there. 03:49 So, these patients will end up with markedly higher levels of circulating cholesterol, which will cause disease. 03:56 So, abnormal LDL, low-density lipoprotein, and intermediate-density lipoprotein transport, and intracellular targeting is how we have this disease. 04:09 And that receptor also takes up IDL. 04:12 There are multiple different mutations, there are many different families that are out there in the world. 04:17 Interestingly, one in 500 of us walking around are heterozygotes for this. 04:25 We have one mutant form of the LDL receptor that came from either mom or dad. 04:29 Being a heterozygote, we have two to three-fold elevated cholesterol. 04:34 Wow. I mean, higher than normal, and it's not because we're eating eggs and fatty foods. 04:41 It's because we don't have the signaling pathway that turn off cholesterol synthesis or clear LDL. 04:48 And if you're a homozygote, you get a mutant form from both mom and dad. 04:52 You have five to six-fold elevated cholesterols. That's not good. 04:57 In fact, the consequences are is that with that elevated circulating cholesterol, you will have accelerated atherosclerosis. 05:04 In a typical homozygote, we'll have a myocardial infarct, that's MI, a heart attack in their 20s due to such aggressive rapid production of atherosclerotic plaque. 05:17 So, that's one consequence. 05:20 Another one is that you will get accumulation of pure cholesterol. 05:23 These nodules on the hands and in other joints within the body are called xanthomas. 05:28 They are not particularly pathologic. 05:32 They don't necessarily cause pain, but they're not particularly attractive. 05:36 They are representation of just pure cholesterol deposition. 05:40 You can also get accumulation of cholesterol in the lose connective tissue around the eyes called xanthelasmas. 05:47 So, these are various manifestations. 05:49 Clearly, the one that's gonna be most important is having your first heart attack when you're in your 20s.
About the Lecture
The lecture Movement of Larger Molecules Across Membranes: Pinocytosis by Richard Mitchell, MD, PhD is from the course Cellular Housekeeping Functions.
Included Quiz Questions
What is the primary etiology of familial hypercholesterolemia?
- LDL receptor dysfunction
- Increased cholesterol synthesis
- Loss of feedback inhibition
- Decreased production of lysosomal enzymes
- Increased LDL absorption from the gastrointestinal tract
Which of the following is taken in by the cells through receptor-mediated endocytosis?
- Low-density lipoprotein
- Folate
- High-density lipoprotein
- Sodium ions
- Oxygen molecules
Which statement best describes receptor-mediated endocytosis?
- It is regulated through feedback inhibition.
- It occurs at the lipid rafts.
- It is a class of caveolin-dependent endocytosis.
- It plays a defensive role against microbial pathogens.
- It plays a primary role in removing cellular debris.
Which of the following is a clinical feature of homozygous familial hypercholesterolemia?
- Xanthomas
- Accelerated myocardial infarction in patients > 30 years old
- Cholesterol is elevated 2-3 fold
- Xanthogranulomatous pyelonephritis
- Periorbital cellulitis
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |