Playlist
Show Playlist
Hide Playlist
Membrane Proteins
-
Slides Cellular Pathology - Interface with the Outside World.pdf
-
Reference List Pathology.pdf
-
Download Lecture Overview
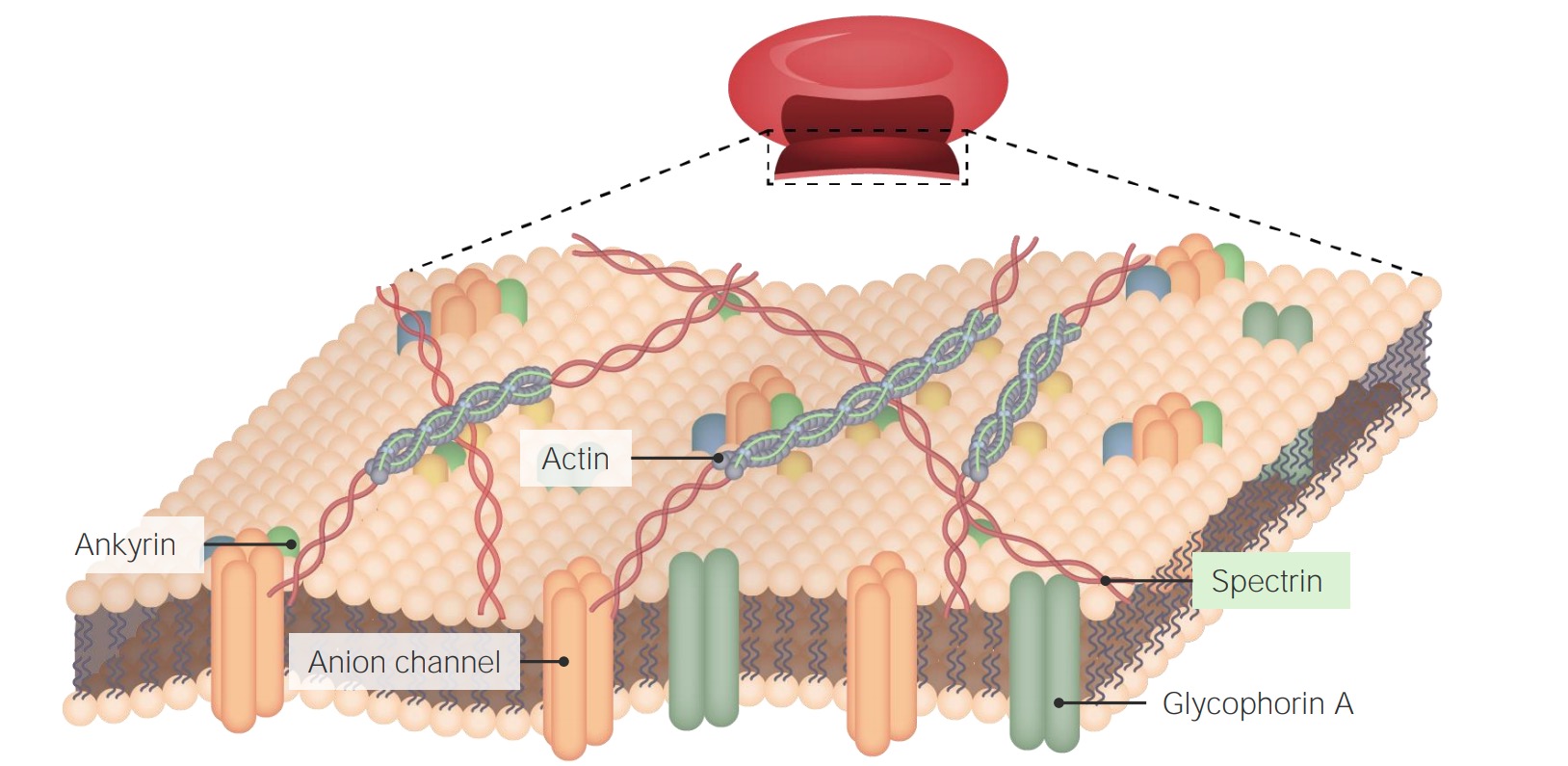
00:01 Alright, let's give the membrane some more functionality besides the lipids and the glycolipids. 00:06 Let's add some proteins. Now, we're talking. 00:08 And membranes are great, but they really can't do a whole lot without something else interacting with them or going through them. 00:17 So, we're gonna add some more labels as we go along here, but on the right lower corner, it says phospholipid molecule. 00:23 Yeah, we're looking at part of a phospholipid bilayer. Okay? And there are a bunch of things sticking through it and out of it. 00:29 So, the cholesterol, we've already previously seen as kind of intersperse throughout the lipid bilayer. That's fine. That's an old friend now. 00:39 But we have integral proteins and based on the lipid composition, the based on cholesterol, based on an array of things, we can get transmembrane proteins. 00:49 These would've been synthesize in the rough endoplasmic reticulum and then stuck as they were translated through in that area. 00:56 And now, we have a transmembrane protein. 00:58 An integral protein that spans from one side to the other. 01:01 Hey, we can do some interesting things with that. 01:03 So, for example, this -- usually, proteins that go through the membrane go through the membrane as an alpha-helix, that's why it's drawn that way, but the outside can interact with cytoskeleton or with another cell. 01:16 In the inside, it can interact with other elements, organelles, and things within the cytosol of the cell. 01:24 So, this allows us now to start doing some pretty interesting things, so we can have communication. We can have channels. 01:32 We can have proteins that get together, that are transmembrane, that allow molecules to go from one side of the membrane to the other side. 01:39 And in a subsequent topic discussion, we will talk about that transport, how we get things from one side to the other side. 01:45 So, we can get channels or carriers that form pores just like you see there that allow things to move from one side to another. 01:53 So, those are the transmembrane proteins. We can have glycoproteins. 01:58 In the same way that we have glycolipids, we can have proteins on the surface that have sugar attached to them. 02:05 Various sugars to make the glyco component of a glycoprotein, and that allows interactions with proteins that bind sugars or lectins. 02:14 So, this allows interaction. 02:16 Importantly, sperm and egg interaction, for example, involves specific glycoproteins on the sperm interacting with receptors on the egg. 02:27 So, we -- none of us would be here without glycoproteins. 02:30 There you go. You can also have peripheral proteins, proteins that don't go through the membrane that are just in one side or the other that are held there because of their interactions with the various lipids or held there because of their interactions with either the cytoskeleton on the inside or the extracytoplasmic matrix on the outside. 02:51 So, those peripheral proteins can also provide other important structural features as we'll see in a minute. 02:58 Just showing here filaments of the cytoskeleton interacting with those peripheral proteins on the inner phase of the membrane. 03:05 And you can also have extracellular matrix interacting with the outside. 03:09 The final element that we're gonna talk about because there's an interesting disease associated with it are glycosylphosphatidylinositol-linked proteins. 03:18 It's pain in the neck to say glycosylphosphatidylinositol, so I'll call it GPI. GPI-linked proteins. 03:24 Okay, so you've probably never heard of that. 03:28 Why do we care about that? All right, so here's some more functionality. 03:32 We have a glycolipid with a phosphate head group, and then we have a GPI. 03:39 So, this is glycosylphosphatidylinositol, phosphatidylinositol, linked through some sugars to a protein. 03:48 So, this protein is not even in the membrane, it's not associated with other membrane proteins, t's just sitting there linked through GPI linkage. 03:56 Another way that we can attach proteins to the surface, but these are kind of interesting because there's a disease associated with failure to make that GPI linkage.
About the Lecture
The lecture Membrane Proteins by Richard Mitchell, MD, PhD is from the course Cellular Housekeeping Functions.
Included Quiz Questions
Which of the membrane proteins is involved in the sperm-egg interaction?
- Glycoproteins
- Channel proteins
- Peripheral proteins
- Integral proteins
- Alpha-helix proteins
Which of the following statements are true regarding glycosylphosphatidylinositol (GPI)-linked proteins?
- They are linked to glycosylphosphatidylinositol (GPI) through a carbohydrate molecule.
- They regulate the movement of ions and proteins across the cell membrane.
- They are formed of hydrophobic regions and hydrophilic regions.
- They have intracellular, transmembrane, and extracellular domains.
- They are involved in maintaining cytoskeletal stability.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Dr. Mitchell makes it amazingly easy to understand this hard-to-grasp concepts!
Dr Richard's enthusiasm and passion for his subject is simply contagious.