Playlist
Show Playlist
Hide Playlist
Hypoplastic Left Heart Syndrome (HLHS)
-
Slides Obstructive heart disease and detour defects.pdf
-
Reference List Pediatric Nursing.pdf
-
Download Lecture Overview
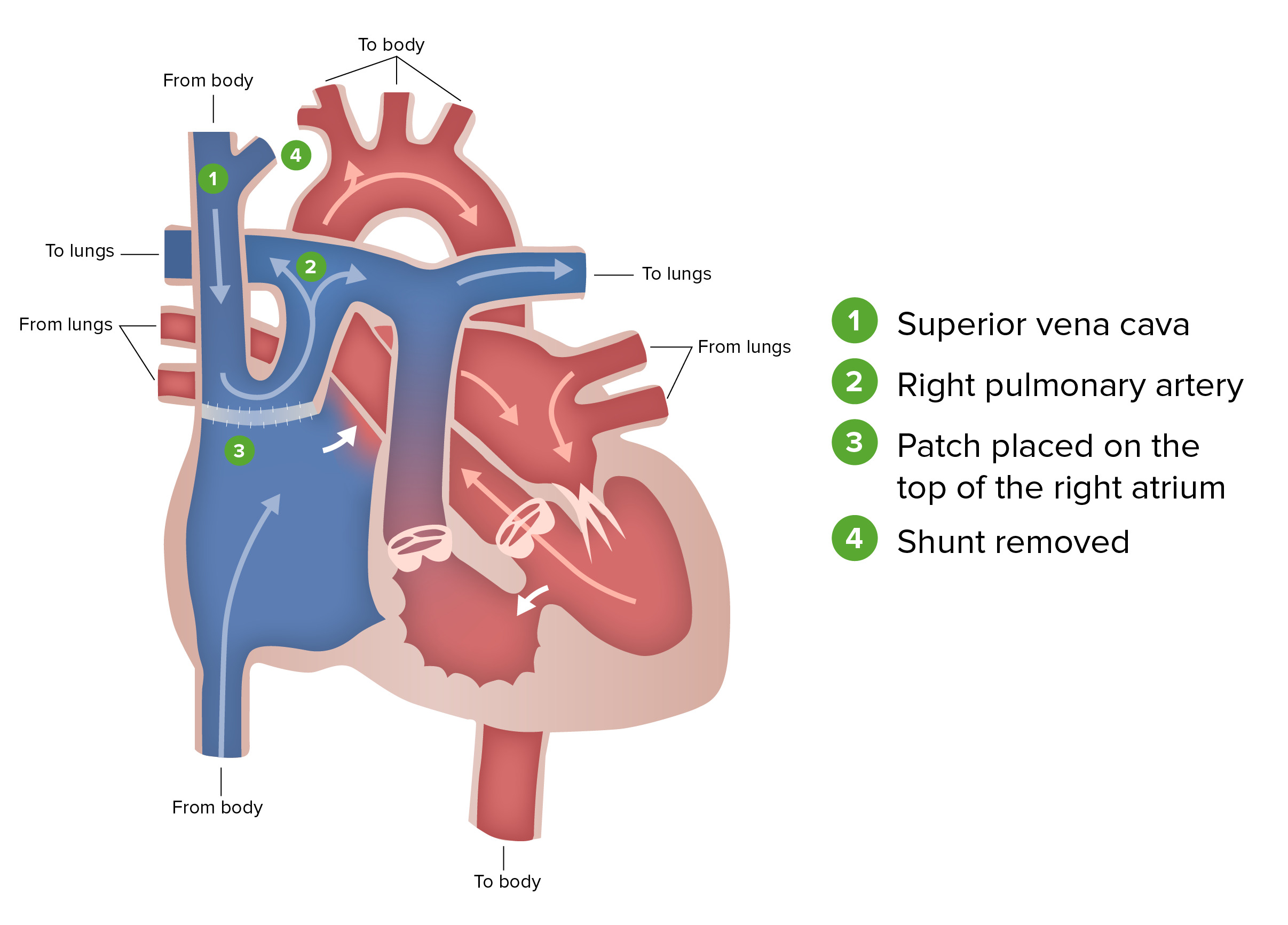
00:01 In this lecture, we're going to discuss obstructive heart disease and detour defects. 00:07 We've already had another talk on cyanotic heart disease. 00:11 So we're talking about congenital heart disease in children. 00:14 Particularly here, we're going to be talking about diseases where the blood is having a hard time getting out of one side of the heart or the other, or when there's a mixing lesion such that blood is going from the left side to the right side. 00:28 Let's start with obstructive congenital heart disease. 00:32 This is the family of heart diseases where the blood is having a hard time getting out of one of the ventricles. 00:39 The classic syndrome that people think of when they hear about this is hypoplastic left heart syndrome. 00:45 This is a high-yield area on exams. 00:49 Generally in hypoplastic left heart syndrome, you have a very small left ventricle. 00:56 It's almost like it isn't there. 00:58 Therefore, the mitral valve is stenotic or atretic. 01:03 The blood is having a hard time getting into that very small left ventricle. 01:07 The aortic valve is also stenotic and atretic, and then the ascending aorta - because there's so little blood going through it - is hypoplastic. 01:19 It's a tiny thread of an aorta. 01:21 The problem is really mostly on the left side of the heart. 01:26 In patients with hypoplastic left heart syndrome, they are going to appear cyanotic at birth - that's because mixed blood is going out to the body, but this is not one of the cyanotic heart disease classified conditions. 01:43 They will be in respiratory distress and this is typically from overcirculating pulmonary blood - more blood keeps going back through that right side and the lungs become congested. 01:54 They may have shock or hypotension, especially if the PDA closes. 02:00 Remember, that patent ductus arteriosus is how the blood is getting to the body. 02:06 If you remember on that left side, there really wasn't much blood going up through that aorta. 02:13 The blood is getting to the body by going through the right side, up into the pulmonary arteries, and then crossing over that patent ductus arteriosus. 02:22 If that closes, now you're having a hard time getting blood to your body and you'll become hypotensive. 02:31 In these patients, they may not have a murmur. 02:35 The appearance of this child will be a cyanotic infant who's in respiratory distress and is hypotensive, but you might not hear a murmur. 02:44 If you listen very carefully, you may hear just a single loud pulmonary valve closing, or a single S2. 02:52 You wouldn't hear a split because that aortic valve really isn't doing much of anything. 02:59 They will often have a hyperactive precordium and you can palpate that right ventricle beating through the chest wall. 03:06 An x-ray will show a very large right heart border. 03:12 As you can see on the slide here, that right side is getting huge over circulation. 03:19 The left side is really not getting much blood at all, so that right atrium and right ventricle will appear enlarged and will bulge into the right side of the x-ray, so you'll see a large right heart border. 03:32 Also, because of this pulmonary overcirculation, you'll see pulmonary congestions and prominent blood vessels on the x-ray. 03:40 But an x-ray really isn't how you're going to make the diagnosis. 03:43 You're going to make the diagnosis through an immediate cardiac echo. 03:48 Any infant who's born tachypnic, cyanotic, is going to get an echo as fast as you can, and that's really how you're going to make this diagnosis. 03:58 If you suspect hypoplastic left heart syndrome and there's going to be a delay in the diagnosis, you may need to intervene before you actually make that diagnosis. 04:12 The first thing we're going to do is start prostaglandins. 04:16 Remember, prostaglandins prevent the closure of a patent ductus arteriosus. 04:23 If that ductus arteriosus closes, that baby is going to become hypotensive and you're going to have end-artery perfusion problems, tissue damage, brain damage, renal damage, liver damage. 04:38 Keeping that patent ductus arteriosus open is critical. 04:43 You're going to watch out for apnea after giving the prostaglandins because that's a side effect of prostaglandins. 04:50 Oftentimes, you maybe intubating simultaneously with giving the prostaglandins. 04:56 When you intubate, you will often need to correct for acidosis. 05:01 Remember, as that blood is not getting to the tissues, lactic acid will build up and we have to correct for that acidosis immediately through both bicarbonate infusions, but also through ventilation. 05:14 Also, blood support is often needed. 05:17 These infants may end up on multiple infusions of both prostaglandins, bicarbonate, and dopamine - and these are all providing life-sustaining features while you await a surgical correction to this condition. 05:32 What is the surgical repair? This is a similar surgical repair to tricuspid atresia, which you'll learn about in the cyanotic heart disease lecture, but it's basically a 3-stage repair that's going to occur over the first several years of the child's life. 05:49 Immediately, birth is the first stage. 05:52 A Blalock-Taussig shunt is seen on your slide in green and it's really a connection between the pulmonary artery and the aorta. 06:02 It's a connection between the left and right side. 06:05 If you will, it's a way of making sort of a permanent patent ductus arteriosus that's allowing systemic blood flow off of that pulmonary artery. 06:16 Also, they will often do an atrial septostomy, which is to literally rip a hole in the septum between the two atrias so you can allow for further mixing. 06:27 This happens essentially within a day or two of birth. 06:31 After about a year, or a few months anyway, we'll proceed to the second stage of the repair, which is called the hemi-Fontan or the Glenn procedure. 06:43 In this procedure, the superior vena cava is hooked up directly with the pulmonary artery. 06:49 Blood from the head is returning and just passively filtering into the lungs, never even sees the heart. 06:55 The blood from the inferior area is still going into the heart. 07:00 At maybe a year or two, or three years of age, they'll complete this 3-stage repair with the Fontan procedure. 07:08 With the Fontan, the inferior vena cava is now also hooked up to the pulmonary artery. 07:14 Now after completion of these three stages, we have a child who has all of the blood returning from the body and passively going to the lungs. 07:24 The blood return, comes back from the lungs, and into that right ventricle, which is now supplying entirely blood through the reconstructed aorta and out to the body. 07:36 Your right ventricle is now powering the systemic blood pressure and pulmonary blood flow is completely passive coming back from the body. 07:48 A number of problems can happen. 07:50 First, pressures can increase on that right side. 07:55 That passive blood flow may result in the lungs forming more high-pressure system, which can cause problems - -- as you'd imagine, you'd see some hepatomegaly in these children as they get older. 08:08 Sometimes to prevent too much pressure, surgeons will fenestrate the Fontan and allow for a little blood to pop off into the heart. 08:19 Another problem is that, right ventricle really isn't designed for lifetime systemic pressures. 08:26 In very rare cases, the patient may also has situs inversus and it's actually a right-sided ventricle that is the left-sided hypoplastic heart. 08:37 This is confusing and there's a lot of variability. 08:40 But the point is that, if the patient - like most children with this condition - has that right heart powering their systemic blood pressure over time, eventually they may be at risk for right-sided heart failure, which is really, in their case, left-sided heart failure. 08:57 This means that, over time, they're going to start getting heart failure and they may require a transplant. 09:02 Most children, if they can make it through the three stages, are going to encounter this around the age of 18 years of age. 09:09 But again, there's a lot of variability.
About the Lecture
The lecture Hypoplastic Left Heart Syndrome (HLHS) by Brian Alverson, MD is from the course Pediatric Cardiology.
Included Quiz Questions
A baby is born with hypoplastic left heart syndrome and has a pulse oximetry reading of 75%. Which of the following is the most important initial therapy?
- Prostaglandin E1
- Bicarbonate
- Oxygen
- Epinephrine
- Dopamine
Which of the following is most likely to cause shock in a newborn with hypoplastic left heart syndrome?
- Closed ductus arteriosus
- Pneumonia
- Administration of prostaglandin E1
- Breastfeeding
- Prematurity
Which of the following is most appropriate initial diagnostic step in a newborn with cyanosis, tachycardia, and heart murmur?
- Echocardiography
- Electrocardiography
- Chest X-ray
- Cardiac catheterization
- Carotid duplex ultrasound
Which of the following may occur as a side effect of prostaglandin administration in patients with cyanotic heart disease?
- Apnea
- Respiratory Infection
- Gastric Reflux
- Hypercoagulability
- Constipation
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Awesome lecture as always! I've been more into my Paediatrics course right now thanks to Dr. Brian, I can understand really fast and easily the concepts and then review more in depth material like books and articles. I love the notes new feature, the slides in PDF and the articles posted here! I really enjoy going through these topics!
Great lecture! As usual. This lecture helped me a lot to improve my understanding of the congenital heart problems in general. The explanations on the surgeries are top notch and on the treatment as well. Thank you!