Playlist
Show Playlist
Hide Playlist
Folate Metabolism Overview and Health
-
Slides VitaminsK,E,B12,FolateReady Biochemistry.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
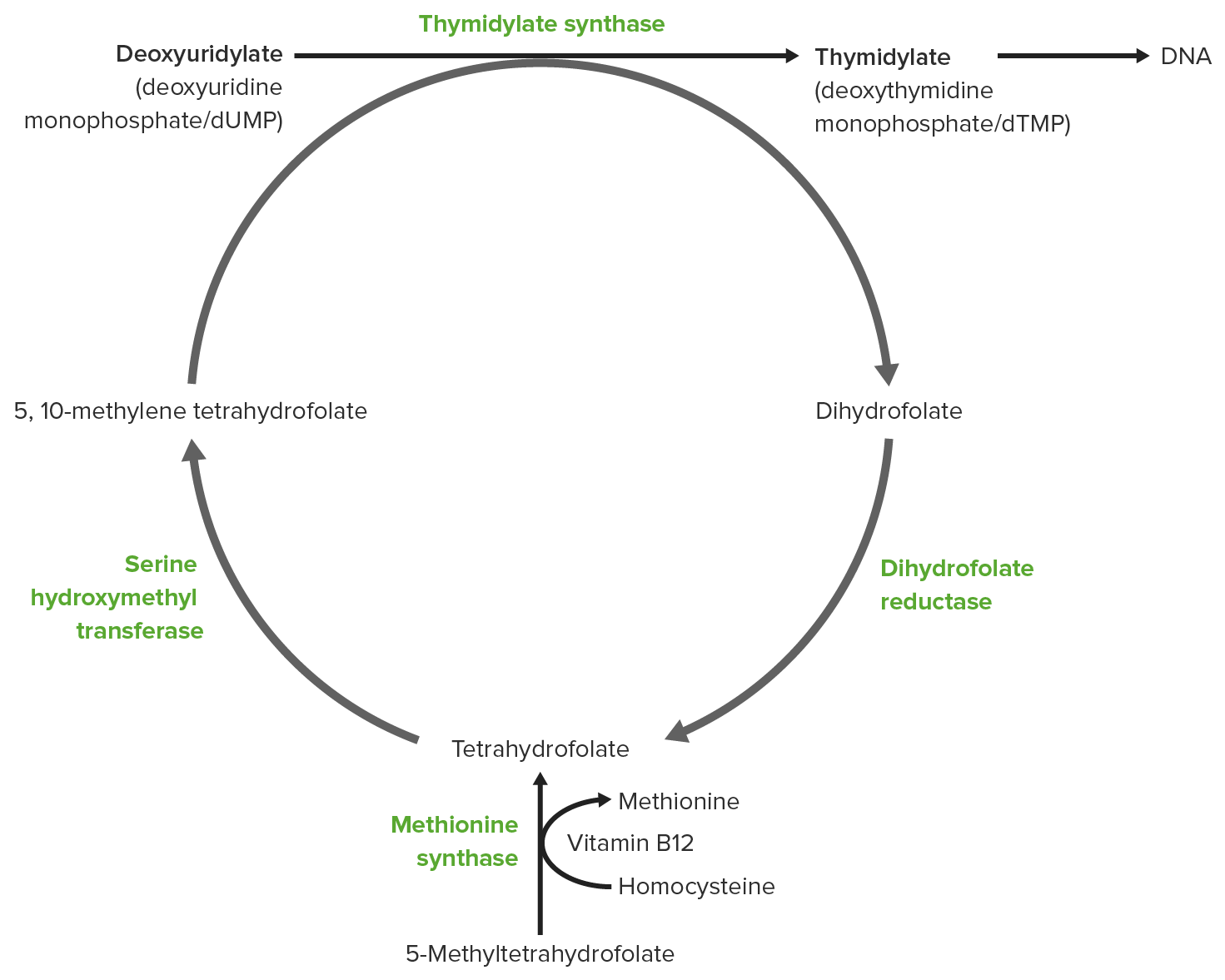
00:01 So I said folate is can sort of be recycled among each other and now I want to spend some time talking about how that recycling process between different folate molecules actually occurs. 00:12 We start this process looking at the very bottom of the screen where we start with the tetrahydrofolate that we’ve been talking about so far. 00:19 Tetrahydrofolate can be converted into the methylene form, the N5, N10 methylene form that I described earlier via the serine/glycine metabolism, reactions that we saw earlier. 00:32 These reactions add in the direction of the methylene. 00:35 They add methylene group between those two nitrogens as we saw before by converting serine to glycine. 00:41 The methylene form can be converted into a methyl form by reduction as we can see in the next slide and that reduction requires electrons from NADPH. 00:51 That produces the N5 methyl group. 00:54 We can see that we now have a methyl group in place of the methylene group that we had before. 01:00 The methionine synthesis, of course, uses this N5 methyl THF to be able to produce the THF that we saw earlier. 01:08 And this will be closed to the cycle on that round of the folate molecules. 01:15 The N5, N10 methylenetetrahydrofolate can also be converted into a group of other folates as we can see here. 01:22 That happens if it gets oxidized. 01:24 So oxidation of that methylene group, donating electrons into NADP to make NADPH produces this N5, N10 methenyltetrahydrofolate or THF as you can see on the screen. 01:38 This molecule has now this carbon double bonded to the nitrogen as you can see. 01:44 The restructuring of that molecule produces another folate known as the N5 formyl tetrahydrofolate as shown and the formyl group is shown in the blue box contrasted with the methenyl group that we’ve shown above. 01:56 So tetrahydrofolate can also be converted into N5 formyl tetrahydrofolate by adding a formyl group that comes from an amino acid metabolism of histidine. 02:06 Histidine is converted into 4-amino glutamic acid during its breakdown process and that 4-amino glutamic acid is the source of the formyl group in making the N5-formyl tetrahydrofolate. 02:18 Well that’s a lot different things that are happening, but those aren’t even the only ones that happened with the N5, N10 methenyltetrahydrofolate. 02:26 The imino group -- that is imino, I-M-I-N-O group -- can be swapped as we see in the reaction moving from the N5,N10 methenyltetrahydrofolate into the N5 4-amino tetrahydrofolate as shown here. 02:40 And we see again a slight difference in configuration of the carbon attached to those two nitrogen molecules. 02:46 We also see, of course, that the tetrahydrofolate can be formiminated but using formyl glutamic acid and producing glutamic acid to produce the N5 4-amino tetrahydrofolate. 02:58 And last but not the least, we see the N5-N10 methenyltetrahydrofolate can be hydrolyzed and lose its group as we see here forming the N10 formyl tetrahydrofolate and the alternate pathway to get into that is from tetrahydrofolate which combines with the formate and ATP to add the formate group as we can see in the very last reaction shown here. 03:23 So folate as I said earlier is important in the diet, it’s important for human health. 03:28 It can’t be made by humans and therefore the only source we have is in our diet. 03:32 Like the other B vitamins, folates are important co-enzymes or small molecules that are not amino acids attached to enzymes that help the enzymes to perform their catalytic function. 03:44 In the late 20th century, it was discovered that deficiencies of folate in pregnant mothers led to neural tube defects in infants. 03:51 And it was that discovery that led to the supplementation of folates in food sources or food stuffs like breads. 04:00 Deficiency of folate is also associated with the variety in other health problems including heart palpitations, damage to nerves, shortness of breath, sore tongues, and other things associated with the mouth and peptic ulcers. 04:16 Deficiencies of folate can also lead to mental problems including confusion, forgetfulness, depression and behavioral disorders. 04:25 Well, we’ve covered a lot of territory here, both the fat-soluble vitamins K and E and the two most important sets of molecules for the movement and for the source of single carbon atoms in biochemical processes.
About the Lecture
The lecture Folate Metabolism Overview and Health by Kevin Ahern, PhD is from the course Vitamins. It contains the following chapters:
- Folate Metabolism Overview
- Folate and Health
Included Quiz Questions
Which of the following is NOT correctly matched regarding folate metabolism?
- THF → N5 - formyl THF — via glycine/serine metabolism
- THF → N5, N10 - methylene THF — via glycine/serine metabolism
- N5 - methyl THF → THF — via methionine synthesis from homocysteine
- THF → N10 formyl THF — via combination with formate
- N5, N10 – methenyl THF → N10 - formyl THF — via hydrolysis reaction
Which of the following is associated with folate deficiency?
- Neural tube defects
- Subdermal hematoma
- Retinal hemorrhage
- Lymphedema
- Tongue infection
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |