Playlist
Show Playlist
Hide Playlist
Epigenetic Alterations and Loss of Proteostasis
-
Slides 01 CellularAging Aging.pdf
-
Download Lecture Overview
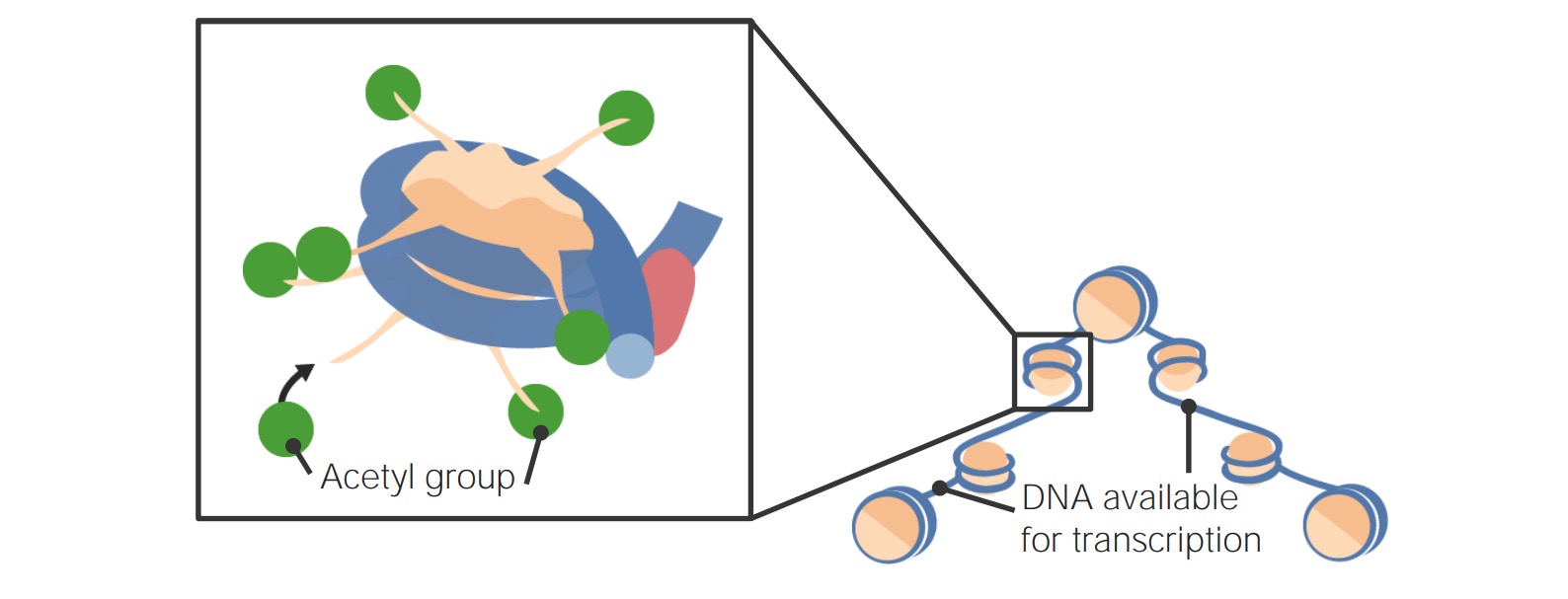
00:01 Epigenetic alterations, we’ve spent a lot of time thinking about epigenetics in the genetics courses, so epigenetics again above the genome. Traditionally, we consider epigenetic factors to be heritable changes that are not to the DNA but to the way the DNA is packaged and how it is accessed. 00:25 We know that these epigenetic factors now can change throughout our lives and alter how and when various genes are expressed in the great variety of cells that we have. These changes in chromatin state in essence are regulating the expression of genes. We’ve definitely spoken about epigenetics being one of the factors that regulates gene expression. When we consider epigenetic factors, I think that you could probably write down at least three different epigenetic alterations that could happen. Go ahead and try that out. Then, let’s move forward and look at a quick review of what those are. First of all, we can have histone modifications. Histones are the cores around which DNA is wrapped. Depending on where those histones are located, DNA may be either accessible for transcription or if it’s wound tightly around the histone, inaccessible for gene transcription. 01:38 These histone modifications can include addition of acetyl groups to the tails and changes in the wrapping of the histone tails, how tightly they hold the DNA onto themselves. 01:53 Again, that lends itself to accessibility issues on the DNA. Another thing in epigenetic alterations we consider is how methylation patterns happen. You know that areas that are highly condensed heterochromatin, we have tight packing and again, lack of access to genes by the transcriptional mechanisms. There’s a connection of DNA methylation patterns to the development and progression of Alzheimer’s disease and how certain proteins fold in there. 02:31 We will definitely cover Alzheimer’s disease in a future lecture in a lot more detail but either way, another epigenetic alteration. All of these epigenetic alterations lend themselves to the concept of chromatin remodeling, so remodeling the areas that are condensed versus opened, and thus the regions that are available for transcription and translation. In general, we see decreasing levels of heterochromatin associated with aging. Epigenetic remodeling or chromatin remodeling actually is changed by some environmental stimuli. This is one of the very active fields of research at the moment. How some of these environmental modifications to, let’s say not DNA, the epigenetic factors can actually become heritable. We’re finding that they can. So, that’s a really exciting field, at least to me at the moment. Next, let’s consider loss of proteostasis. 03:44 Clearly, if we have changes in transcription rates and frequencies of a variety of different genes then we have a loss of proteostasis, simply meaning that cells perhaps lose the ability to do quality control on their own protein production. The protein production has gone awry because of these other modifications and insults to the DNA or reactive oxygen species, all of these different things. We see that the loss of proteostasis in beta-amyloid plaques and tau proteins and such that we see in Alzheimer’s and Parkinson’s. We also see the disruption of protein homeostasis in cataracts that’s responsible for the cloudiness that occurs in cataracts, so again, disorders clearly associated with loss of proteostasis or homeostasis of the proteins. 04:42 Things are getting a little bit dysregulated. Accumulation of these damaged proteins as we see in Parkinson’s and Alzheimer’s disease clearly cause some phenotypic effects, even can be toxic if not just interrupting signaling.
About the Lecture
The lecture Epigenetic Alterations and Loss of Proteostasis by Georgina Cornwall, PhD is from the course Aging. It contains the following chapters:
- Epigenetic alterations
- Loss of proteostasis
Included Quiz Questions
Which of the following best describes epigenetic alterations?
- Heritable changes in phenotype without a change in genotype
- Changes in genotype without a change in phenotype
- Heritable changes in both genotype and phenotype
- Changes in DNA without a change in chromatin state
- Non-hereditary changes in both phenotype and genotype
Alzheimer's disease, Parkinson's disease, and cataracts are examples of which cell aging process?
- Loss of proteostasis
- Telomere attrition
- Spontaneous hydrolytic reaction
- Apoptosis
- Accumulation of free radicals in the cell
Which of the following is NOT an example of an epigenetic alteration?
- Changes in nucleotide sequences that were caused by environmental stimuli
- Environmental stimuli that caused chromatin remodeling
- Changes in patterns of methylation of DNA, altering accessibility for gene transcription
- Histone tail modification, altering DNA accessibility for gene transcription
- An inheritable change in the shape of chromatin without changes to genetic material
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |