Playlist
Show Playlist
Hide Playlist
Bohr Model and Atomic Structure
-
Slides ElectronicStructure1 Physics.pdf
-
Download Lecture Overview
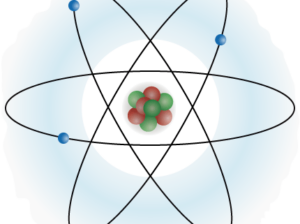
00:02 Nachdem wir nun über den Atomkern gesprochen haben und was sich im Zentrum eines Atoms befindet, sind wir jetzt bereit, die elektronische Struktur eines Atoms zu besprechen. 00:10 Wir beginnen mit dem so genannten Bohrschen Atommodell. 00:13 Und das ist historisch gesehen eine sehr bedeutende Entwicklung, wie wir darüber denken, was ein Atom ist. 00:18 Wie es strukturiert ist und wie es sich daher verhält. 00:22 Historisch gesehen, wussten die Menschen ursprünglich, dass positive und negative Ladungen existieren. 00:26 Und sie hatten dies in Experimenten gesehen. 00:28 Aber sie wussten nicht wirklich, wie sich diese Ladungen im Atom organisieren. 00:31 Im sogenannten Thomsonschen Atommodell (Plumpuddingmodell), haben wir diese Art der Verteilung hier auf der linken Seite. 00:36 Wo wir eine große positive Ladung haben könnten, können wir ein positives Zeichen in der Mitte sehen. 00:41 Das wurde sozusagen auf das ganze Atom verteilt, mit vielleicht negative Ladungen, die sich wie Pflaumen im Pudding verteilen, deshalb Plumpuddingmodell. 00:48 Aber dann wurden einige Experimente durchgeführt, bei denen verschiedene Arten von Teilchen auf ein Atom geschossen wurden, um zu sehen, wie sie daran abprallen. 00:56 Und zur großen Überraschung aller stellten sie fest, dass sie an einem harten Kern im Zentrum des Atoms abprallten, aber sonst alles durchgängig war. 01:03 Und das ist das Bohrsche Modell, die Abbildung, die Sie sehen können auf der rechten Seite. 01:07 In der Mitte haben wir den Kern, den wir bereits besprochen haben. 01:10 Er hat positive Ladungen, die auch Protonen genannt werden als auch neutrale Ladungen, die Neutronen. 01:15 Und auf der Außenseite befinden sich die Elektronen, die sehr klein sind. Kleine Teilchen, die mit sehr großer Geschwindigkeit um den Kern kreisen, in einer, im Verhältnis zum Kern, großen Distanz. 01:24 Dieses Bohr-Atom hat auch noch ein paar weitere Eigenschaften. 01:27 Vor allem und vielleicht am wichtigsten, die elektronische Struktur. 01:31 Diese elektronische Struktur hat auch verschiedene Energieniveaus (Kreise oder Schalen). 01:35 Dies sind die Orte, an denen sich die Elektronen aufhalten dürfen, die um Ihren Kern kreisen. 01:40 Anders als Planeten, die sich gegenseitig umkreisen, können sie die Umlaufbahnen nicht beliebig festlegen. 01:45 Damit Elektronen einen Kern umkreisen können, müssen sie sich in sehr separaten, sehr unterschiedlichen Energieniveaus bewegen. 01:51 Zum Beispiel dieses erste Energieniveau, das dem Kern am nächsten liegt, wird als erstes Energieniveau (1. Schale, K-Schale) bezeichnet oder der Grundzustand dieses Atoms. 01:59 Und dann könnten die Elektronen in das zweite Energieniveau (2. Schale, L-Schale) übergehen und so weiter. 02:03 Aber auch hier gilt, dass sie separat sein müssen. 02:05 Es müssen ganz bestimmte Ebenen sein, und sie können nie zwischen den Ebenen stehen. 02:09 Eine weitere Sache zu diesen Orbitalen, die wir besprechen werden, auch nochmal ein bisschen später, ist, dass es nur eine bestimmte Anzahl von Elektronen in diesen Kreisen gibt. 02:18 Sie sehen also, dass wir bereits viele Einschränkungen besprechen, um zu klären wie dieses Bohrsche Atommodell funktionieren kann. 02:24 Behalten Sie also auch die Einschränkungen im Auge, die wir erörtern: 1. Elektronen können nur in bestimmten Energieniveaus sein. 02:30 Und 2., es kann nur eine bestimmte Anzahl von Elektronen auf einem bestimmten Energieniveau geben. 02:35 Diese Zahl ergibt sich aus 2 mal dem Energieniveau zum Quadrat. 02:39 So zum Beispiel in der ersten Energiestufe, das ist der Grundzustand, der dem Kern am nächsten ist. 02:44 Wir können zwei mal 1 zum Quadrat oder zwei Elektronen in diesem Energieniveau haben. 02:48 Und wir können dies für eine Reihe von anderen Energieniveaus berechnen. 02:51 Zum Beispiel für dieses zweite Energieniveau: Wir haben zwei mal 2 zum Quadrat oder zwei mal 4, was 8 Elektronen ergibt, die auf dem zweiten Energieniveau liegen. 03:00 Das typische Wasserstoffatom, auf das wir uns im weiteren Verlauf fokussieren werden, sieht in etwa so aus. 03:06 In seinem Zentrum befindet sich ein Proton, ohne Neutronen und ohne andere Protonen. 03:10 Nur eine positive Ladung, die genau in der Mitte sitzt. 03:13 Und dann umkreisen ihn die Elektronen. 03:15 Und nochmal, hier gibt es keine weiteren Neutronen irgendwo im Kern. 03:18 Und das niedrigste Energieniveau, das n = 1-Niveau, heißt wiederum "Grundzustand", in dem sich das Elektron befinden muss. 03:25 Der Begriff "Grundzustand" taucht auch immer wieder auf. 03:29 Seien Sie also mit der Tatsache vertraut, dass, wenn ich "Grundzustand" sage, ich mich auf das Energieniveau n = 1 beziehe. 03:34 Wenn die Elektronen in einen höheren Zustand übergehen, wie bei n = 2 oder sogar das Energieniveau n = 3, sagen wir, dass das Elektron oder das Atom selbst in einem angeregten Zustand ist. 03:45 Es hat mehr Energie. 03:46 Denn es braucht Energie, um ein Elektron auf eine höheres Energieniveau zu bringen. 03:51 Zu diesem höheren Zustand: Eine Möglichkeit, ein Elektron auf ein höheres Energieniveau zu bringen, von einem niedrigeren Zustand wie dem Grundzustand, ist, es mit Licht zu bestrahlen. 04:00 Hier sehen wir ein Photon auf dem Weg nach innen. 04:02 Es trifft das Elektron und das Elektron wird angehoben auf das höhere Energieniveau. 04:06 Und dann würden wir wieder sagen, dass sich das Atom in einem angeregten Zustand befindet, wenn sich das Elektron auf einem höheren Energieniveau befindet als dem Grundzustand. 04:15 Wenn das Elektron aus diesem angeregten Zustand zurückkehrt, haben wir ein sehr wichtiges Phänomen, das genau das Gegenteil ist von dem, was ich gerade gesagt habe. 04:23 Anstatt ein Photon zu absorbieren und in eine höhere Energie zu gehen, fällt es auf ein niedrigeres Energieniveau und emittiert Licht. 04:30 Genau das kann man auf dem Bild hier sehen. 04:33 Wir haben ein Elektron im angeregten Zustand. 04:35 Es fällt in den Grundzustand. 04:37 Und es sendet ein Photon aus. 04:39 Und dieses Photon hat eine bestimmte Menge an Energie, wenn es emittiert wird. 04:43 Wir können diese Energie einfach mit Hilfe der Energieerhaltung errechnen. 04:47 Wenn das Atom die Energiemenge 1 hatte und sich dann zu einer geringeren Energiemenge verändert, können wir die Differenz berechnen. 04:53 Wir können berechnen, wie viel Energie verloren gegangen ist. 04:56 Wir schreiben das in der gleichen Terminologie auf, die wir bisher verwendet haben, mit einem Delta-Zeichen zur Darstellung der Veränderung. 05:01 Die Änderung der Energie des Atoms ist auch die Endenergie, die es hat, bzw. die erregte Energie, die Anfangsenergie, die erste Energie, abzüglich der Grundzustandsenergie. 05:13 Und das ist so, weil wir die erregte Energie haben, die eine größere Zahl ist. 05:17 Und dann ist die Energie im Grundzustand eine kleinere Zahl. 05:19 Mit dieser Gleichung können wir herausfinden, welche Energie das Photon haben muss. 05:24 Auch hier gilt der Grundsatz der Energieerhaltung. 05:26 Die Photonenenergie ist eigentlich etwas, das wir schon berechnet haben, falls Sie sich noch daran erinnern. 05:31 Die Photonenenergie hier ist einfach die Plancksche Konstante h mal der Frequenz dieses Photons. 05:37 Diese wiederum bestimmt, welche Farbe das Photon hat. 05:40 Wenn es sich um ein rotes Photon handelt, ist es ein Licht mit niedrigerer Energie, es ist eine niedrigere Frequenz. 05:44 Und umgekehrt für die höhere Frequenz: Es ist blauer bei höheren Frequenzen, die höheren Energien entsprechen. 05:51 Und vergessen Sie auch nicht, dass wir auch dies berechnen können aus der Wellenlänge, wenn wir die Frequenz nicht kennen, die Wellenlänge des Lichts aber bekannt ist. 05:57 Da wir die beiden mit unserer Geschwindigkeitsgleichung in Beziehung setzen können, die wir bereits erörtert haben. 06:03 Diese atomare Struktur kann weitergeführt werden. 06:05 Wir haben bisher nur das erste und das zweite Energieniveau gezeigt. 06:08 Aber wir könnten im Prinzip zu immer höheren Energieniveaus übergehen. 06:11 Und die würden in etwa so aussehen. 06:13 Sie sind nicht gleichmäßig verteilt. 06:14 Das kann man am Atom sehen, vom Zentrum des Atoms zum Grundzustand haben wir ein Radius, eine Entfernung. 06:21 Und dann, wenn wir immer weiter nach oben gehen, nehmen diese Abstände von einem Energieniveau zum nächsten ab.
About the Lecture
The lecture Bohr Model and Atomic Structure by Jared Rovny, PhD is from the course Electronic Structure.
Included Quiz Questions
Before the Bohr model, how were positive charges considered to be distributed in an atom?
- Spread out throughout the atom
- Concentrated in the center
- Orbiting the center
- Forming concentric rings
- Spread uniformly in a spherical shell
What are the possible energy levels n of an electron in an atom? And what is the maximum number of electrons that can occupy each level?
- n = 1, 2, 3, ..., Max. number of electrons at each level : 2n²
- n = 0, 1, 2, ..., Max. number of electrons at each level : n²
- n = -2, -1, 0, 1, 2, ..., Max. number of electrons at each level : 2n²
- n = 1/2, 2/2, 3/2, ..., Max. number of electrons at each level : n²
- n = 0, 1/2, 2/2, 3/2, ..., Max. number of electrons at each level : 2n²
How many electrons can occupy the n = 3 energy level?
- 18
- 16
- 20
- 14
- 10
How is a photon emitted from an excited atom? And what is the relation between the emitted photon energy E and its frequency f?
- When electrons in an atom are dropped from an excited state to a lower energy state the energy difference is released in the form of the emission of a photon, E = hf
- By raising electrons to higher energy levels, E = hf
- By swapping two electrons in adjacent energy levels, E = E₀ f/f₀
- By losing an electron from the atom, E = E₀ f/f₀
- The excited atom collides with other atoms and releases its kinetic energy in the form of an emitted photon, E = hf
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |