Amino acids Amino acids Organic compounds that generally contain an amino (-NH2) and a carboxyl (-COOH) group. Twenty alpha-amino acids are the subunits which are polymerized to form proteins. Basics of Amino Acids (AAs) can be acquired through the breakdown of intracellular or ingested dietary proteins Proteins Linear polypeptides that are synthesized on ribosomes and may be further modified, crosslinked, cleaved, or assembled into complex proteins with several subunits. The specific sequence of amino acids determines the shape the polypeptide will take, during protein folding, and the function of the protein. Energy Homeostasis. Amino acids Amino acids Organic compounds that generally contain an amino (-NH2) and a carboxyl (-COOH) group. Twenty alpha-amino acids are the subunits which are polymerized to form proteins. Basics of Amino Acids can enter 3 metabolic routes within the body. They can 1) be recycled to synthesize new proteins Proteins Linear polypeptides that are synthesized on ribosomes and may be further modified, crosslinked, cleaved, or assembled into complex proteins with several subunits. The specific sequence of amino acids determines the shape the polypeptide will take, during protein folding, and the function of the protein. Energy Homeostasis; 2) combine with cofactors and substances to create amino acid Amino acid Amino acids (AAs) are composed of a central carbon atom attached to a carboxyl group, an amino group, a hydrogen atom, and a side chain (R group). Basics of Amino Acids derivatives; or 3) be catabolized into their functional groups and carbon skeletons. This process releases ammonium, which moves into the urea Urea A compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids. Urea Cycle cycle and produces intermediates for energetic metabolic pathways.
Last updated: Feb 17, 2023
Amino acids Amino acids Organic compounds that generally contain an amino (-NH2) and a carboxyl (-COOH) group. Twenty alpha-amino acids are the subunits which are polymerized to form proteins. Basics of Amino Acids (AAs) follow 3 main metabolic pathways for their metabolism:
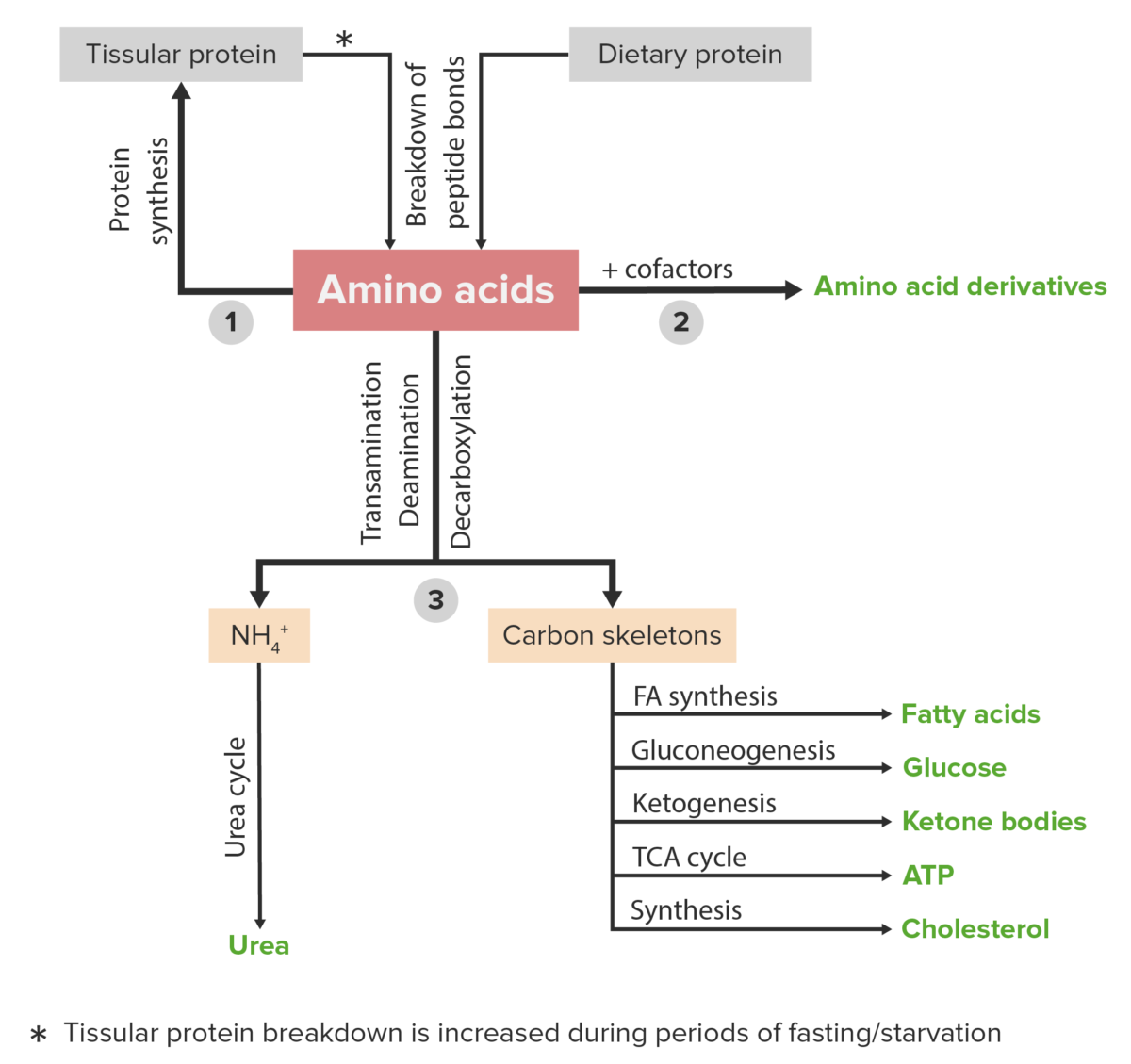
Schematic diagram of the metabolism of amino acids, including the 3 major pathways: reutilization in the synthesis of new proteins, union with cofactors to produce amino acid derivatives, and catabolism. Catabolism of amino acids includes the removal of functional groups and the breakdown of the carbon skeletons.
Amino acids Amino acids Organic compounds that generally contain an amino (-NH2) and a carboxyl (-COOH) group. Twenty alpha-amino acids are the subunits which are polymerized to form proteins. Basics of Amino Acids can be used to assemble many substances. The image below shows the most important AA-derived substances in humans.
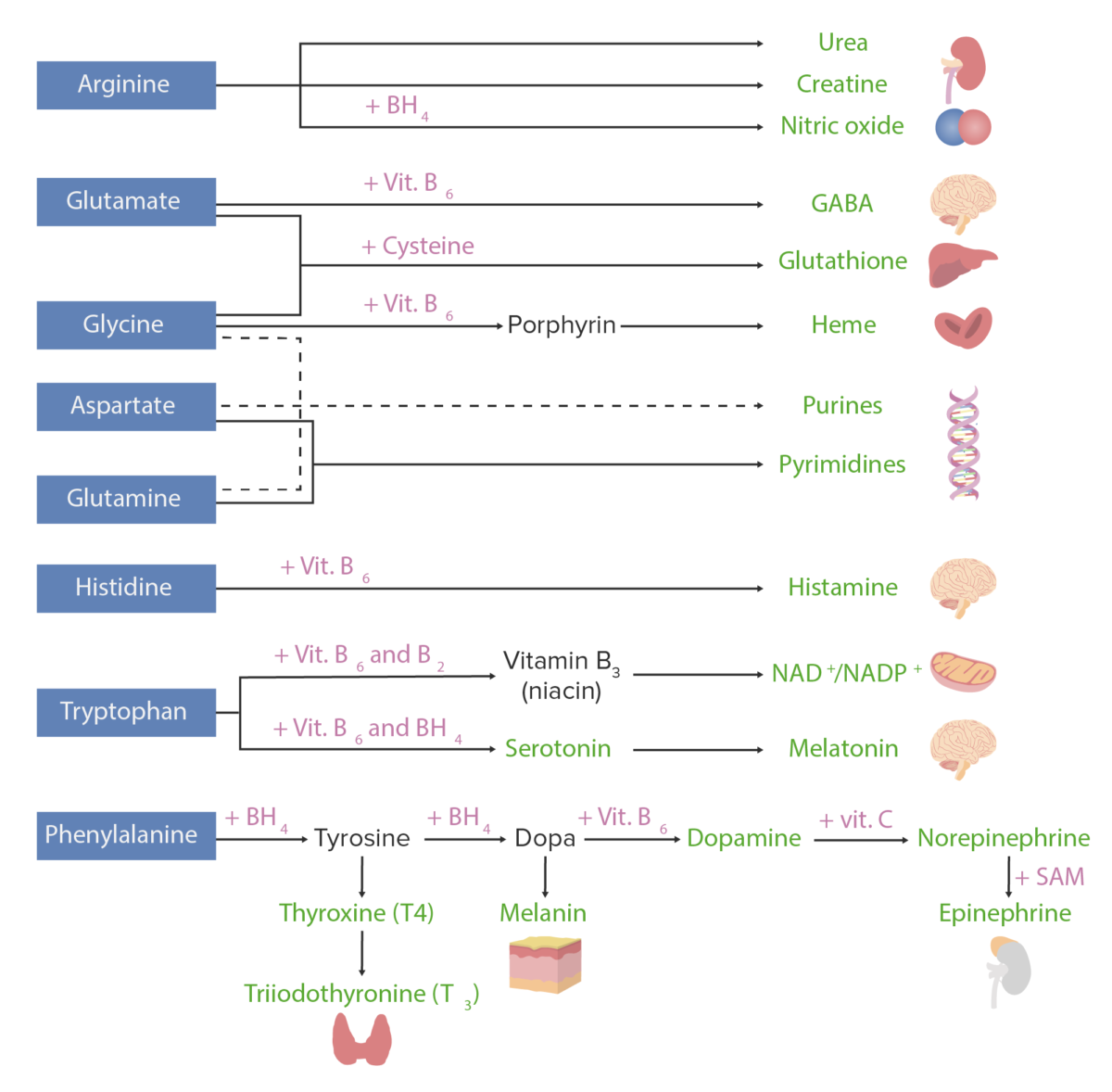
Amino acid derivatives. Amino acids (in blue) are combined with certain cofactors or other substrates (in pink) to make several biologically-important substances (in green).
Image by Lecturio.Transamination is the transfer of an amino group from an alpha-AA to an alpha-keto acid, which is an AA AA Amyloidosis with an alpha-keto group (=O) instead of an alpha-amino group (NH2).
This process is need-dependent. If there is an excess of a type of AA AA Amyloidosis, the amino group of that type can be transferred to make other types of AAs that the body currently needs.
All of the common AAs participate in transamination, except lysine, threonine, proline Proline A non-essential amino acid that is synthesized from glutamic acid. It is an essential component of collagen and is important for proper functioning of joints and tendons. Synthesis of Nonessential Amino Acids, and hydroxyproline, which catabolize via a dehydrogenase.
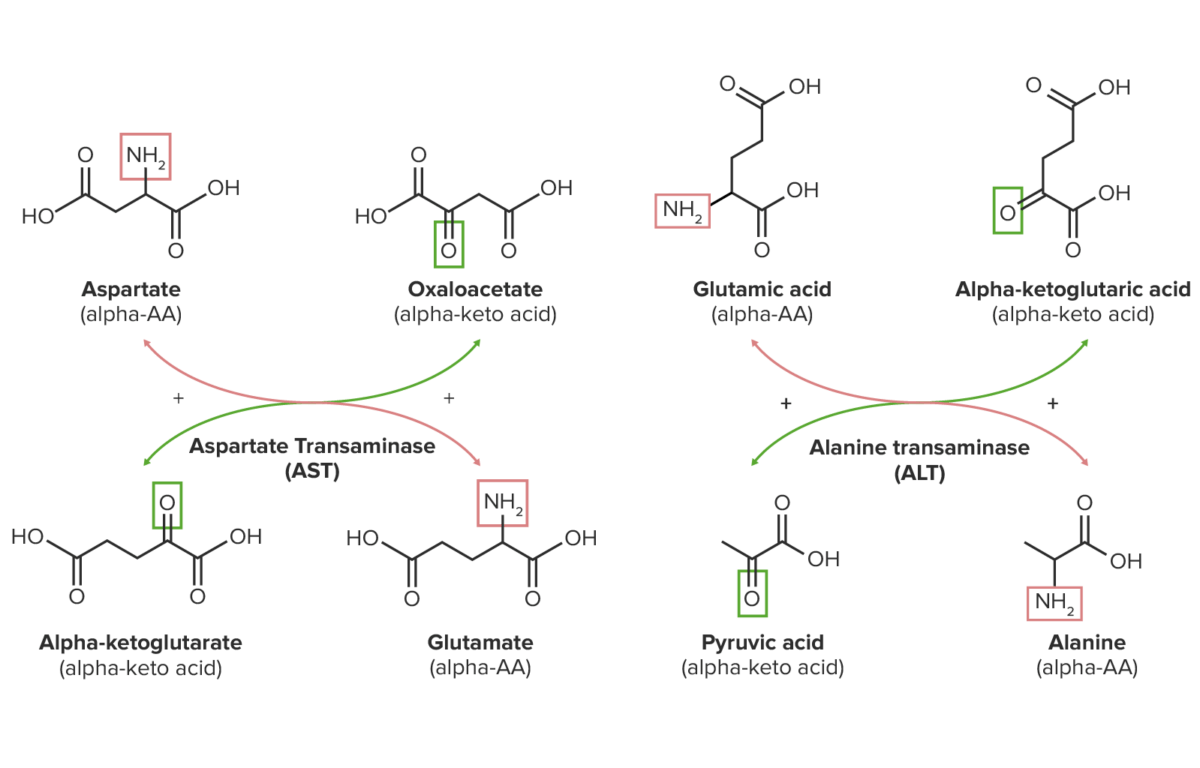
Schematic diagram of the transamination reactions of aspartate and glutamate (glutamic acid):
Amino groups are highlighted in red.
Keto groups are highlighted in green.
Both enzymes Enzymes Enzymes are complex protein biocatalysts that accelerate chemical reactions without being consumed by them. Due to the body’s constant metabolic needs, the absence of enzymes would make life unsustainable, as reactions would occur too slowly without these molecules. Basics of Enzymes catabolize reversible reactions, which are essential for the transport of nitrogen Nitrogen An element with the atomic symbol n, atomic number 7, and atomic weight [14. 00643; 14. 00728]. Nitrogen exists as a diatomic gas and makes up about 78% of the earth’s atmosphere by volume. It is a constituent of proteins and nucleic acids and found in all living cells. Urea Cycle from tissues to the liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy and into the urea Urea A compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids. Urea Cycle cycle.
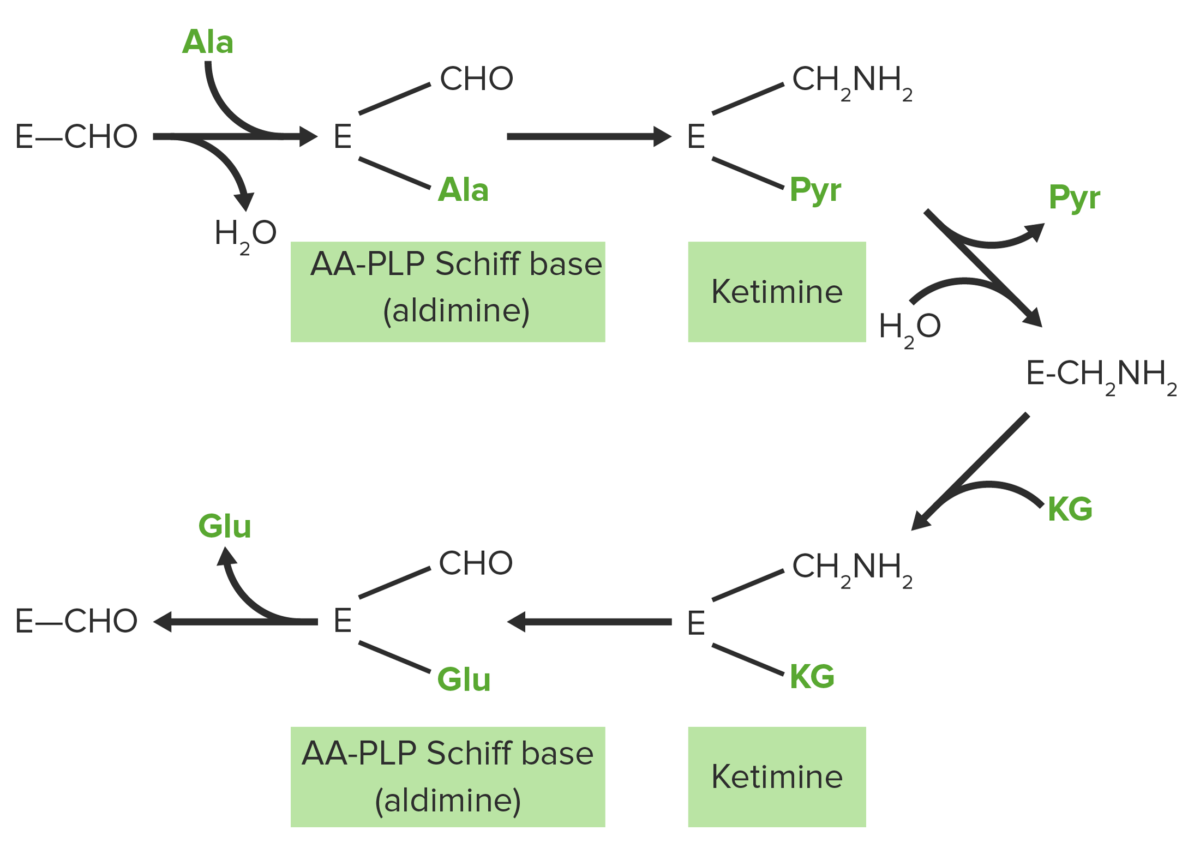
Ping pong bi-bi mechanism of PLP-dependent enzyme-catalyzed transamination. Aminotransferase reaction occurs in 2 stages consisting of 3 steps: transamination, tautomerization, and hydrolysis. In the first stage, the alpha-amino group of the amino acid is transferred to PLP, yielding an alpha ketoacid and pyridoxamine phosphate (PMP). In the second stage of the reaction, the amino group of PMP is transferred to a different alpha-keto acid to yield a new alpha-amino acid and PLP.
Image by Lecturio.Steps:
Deamination is the process through which amino groups are stripped from AAs, releasing free cytotoxic Cytotoxic Parvovirus B19 ammonia Ammonia A colorless alkaline gas. It is formed in the body during decomposition of organic materials during a large number of metabolically important reactions. Note that the aqueous form of ammonia is referred to as ammonium hydroxide. Acid-Base Balance: ammonia Ammonia A colorless alkaline gas. It is formed in the body during decomposition of organic materials during a large number of metabolically important reactions. Note that the aqueous form of ammonia is referred to as ammonium hydroxide. Acid-Base Balance → ammonium → urea Urea A compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids. Urea Cycle or uric acid Uric acid An oxidation product, via xanthine oxidase, of oxypurines such as xanthine and hypoxanthine. It is the final oxidation product of purine catabolism in humans and primates, whereas in most other mammals urate oxidase further oxidizes it to allantoin. Nephrolithiasis via the urea Urea A compound formed in the liver from ammonia produced by the deamination of amino acids. It is the principal end product of protein catabolism and constitutes about one half of the total urinary solids. Urea Cycle cycle in the liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy.
1. Oxidative deamination:
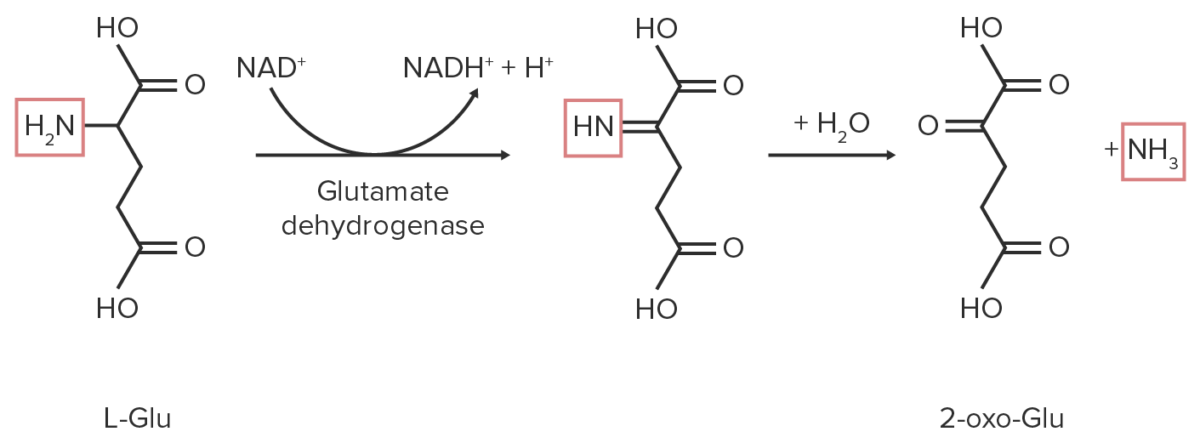
Schematic diagram of the oxidative deamination reaction of glutamate. The nitrogen-containing functional groups are highlighted in red.
Image by Lecturio.2. Hydrolytic deamination:
Water reacts with the amino group, irreversibly attaching an OH group and eliminating the amino group in the form of ammonia Ammonia A colorless alkaline gas. It is formed in the body during decomposition of organic materials during a large number of metabolically important reactions. Note that the aqueous form of ammonia is referred to as ammonium hydroxide. Acid-Base Balance.
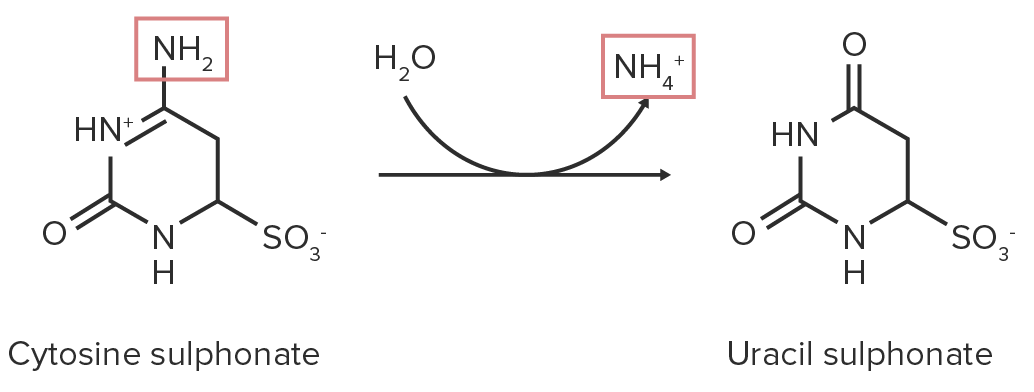
Schematic diagram of a hydrolytic deamination reaction. The nitrogen-containing functional groups are highlighted in red.
Image by Lecturio.3. Eliminative deamination:
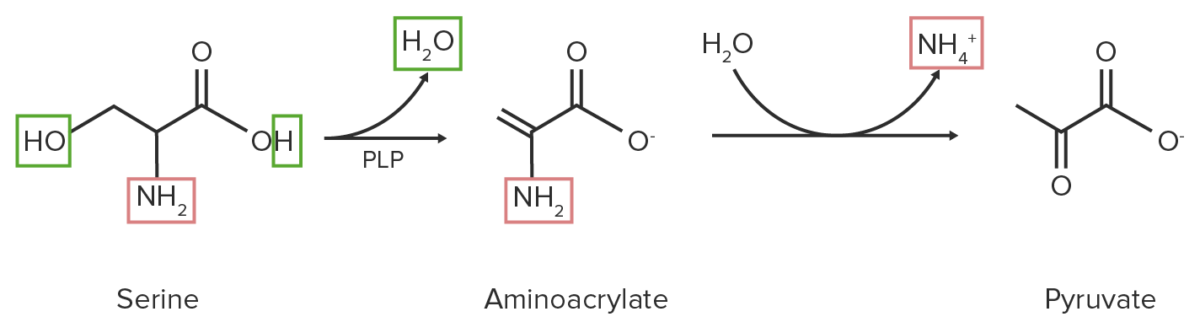
Schematic diagram of the eliminative deamination reaction of serine. The nitrogen-containing functional groups are highlighted in red, while the water molecule (H2O) and its components are highlighted in green.
Image by Lecturio.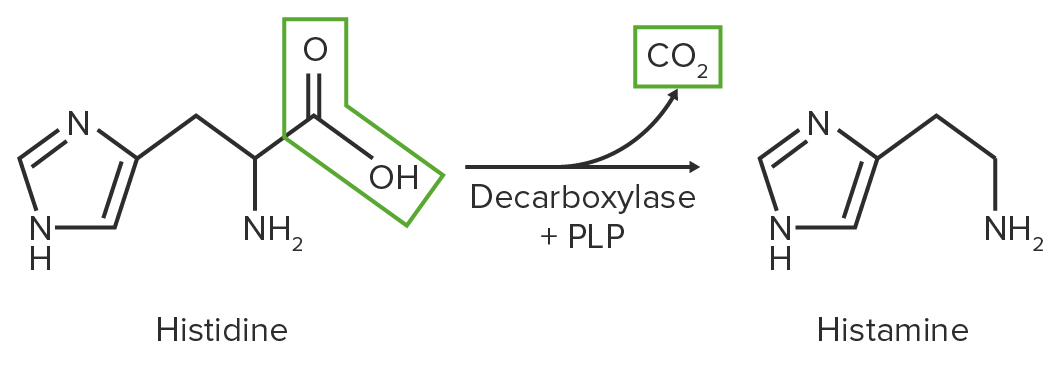
Schematic diagram of the decarboxylation reaction of histidine to histamine
Image by Lecturio.The catabolism of AAs involves anaplerotic reactions (chemical reactions that form intermediates of metabolic pathways).
The breakdown of the carbon skeleton of AAs can be classified by the metabolic pathways to which their catabolic products will serve as intermediates:
| Glucogenic AAs | Ketogenic AAs | Glucogenic/Ketogenic AAs |
|---|---|---|
|
|
|
All AAs are broken down into 1 of 6 intermediates (see green boxes in the images below): pyruvate Pyruvate Derivatives of pyruvic acid, including its salts and esters. Glycolysis, acetyl-CoA Acetyl-CoA Acetyl CoA participates in the biosynthesis of fatty acids and sterols, in the oxidation of fatty acids and in the metabolism of many amino acids. It also acts as a biological acetylating agent. Citric Acid Cycle, oxaloacetate Oxaloacetate Derivatives of oxaloacetic acid. Included under this heading are a broad variety of acid forms, salts, esters, and amides that include a 2-keto-1, 4-carboxy aliphatic structure. Citric Acid Cycle, alpha-ketoglutarate, succinyl-CoA Succinyl-CoA Citric Acid Cycle, or fumarate Fumarate Citric Acid Cycle.
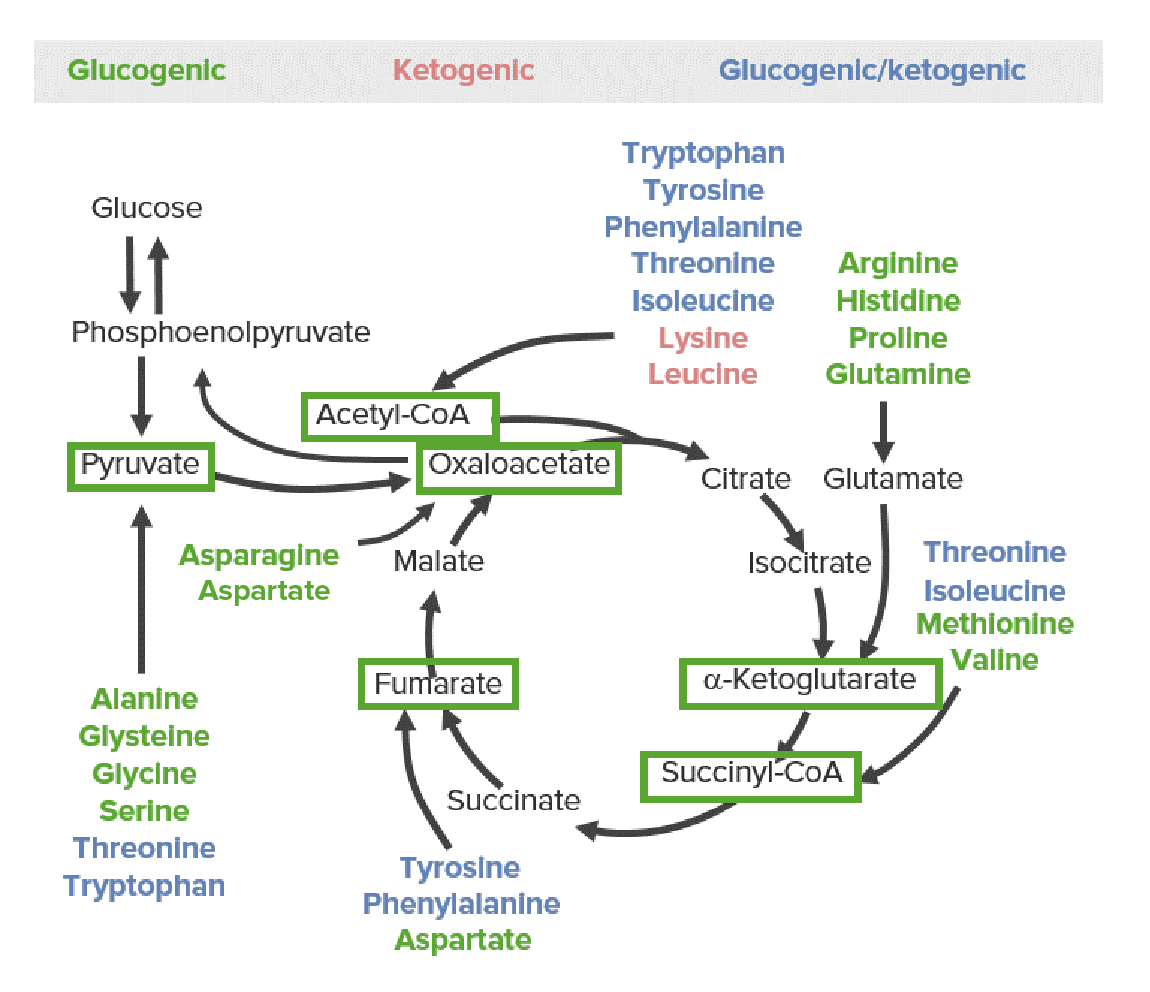
The 3 categories of catabolic products of amino acids: glucogenic (green), ketogenic (red), and both glucogenic and ketogenic (blue). The glucose-pyruvate pathway on the left represents glycolysis and gluconeogenesis. The cyclic pathway on the right represents the citric acid cycle. All amino acids are broken down into 1 of 6 intermediates (green boxes): pyruvate, acetyl-CoA, oxaloacetate, alpha-ketoglutarate, succinyl-CoA, or fumarate.
Glucogenic AAs
Metabolized to pyruvate Pyruvate Derivatives of pyruvic acid, including its salts and esters. Glycolysis or metabolites of the citric acid cycle Citric acid cycle The citric acid cycle, also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle, is a cyclic set of reactions that occurs in the mitochondrial matrix. The TCA cycle is the continuation of any metabolic pathway that produces pyruvate, which is converted into its main substrate, acetyl-CoA. Citric Acid Cycle (CAC):
Catabolic products either move into the CAC to produce energy or are used as substrates for gluconeogenesis Gluconeogenesis Gluconeogenesis is the process of making glucose from noncarbohydrate precursors. This metabolic pathway is more than just a reversal of glycolysis. Gluconeogenesis provides the body with glucose not obtained from food, such as during a fasting period. The production of glucose is critical for organs and cells that cannot use fat for fuel. Gluconeogenesis.
Ketogenic AAs
Metabolized directly to acetyl-CoA Acetyl-CoA Acetyl CoA participates in the biosynthesis of fatty acids and sterols, in the oxidation of fatty acids and in the metabolism of many amino acids. It also acts as a biological acetylating agent. Citric Acid Cycle, then enter 1 of 3 metabolic pathways:
Glucogenic and Ketogenic AAs
Metabolized to intermediates of lipidic as well as glucogenic pathways:
To recall the metabolic pathways of the carbon skeletons of amino acids Amino acids Organic compounds that generally contain an amino (-NH2) and a carboxyl (-COOH) group. Twenty alpha-amino acids are the subunits which are polymerized to form proteins. Basics of Amino Acids, remember:
The following conditions are disorders of amino acid metabolism Disorders of amino acid metabolism Amino acids are the building blocks of proteins, whose production is a tightly regulated enzymatic cascade. Disorders of the enzymes involved in amino acid metabolism are often serious and present early in life. Congenital errors in amino acid metabolism are due to either impaired synthesis or degradation. Disorders of Amino Acid Metabolism. Depending on the country and the individual U.S. state, newborn Newborn An infant during the first 28 days after birth. Physical Examination of the Newborn infants may be routinely screened for these disorders (except for alkaptonuria).